Faculty: Lisa S. Pair, APRN, DNP, WHNP-BC, is Assistant Professor of Nursing at the University of Alabama at Birmingham, School of Nursing, in Birmingham, Alabama.
William E. Somerall, Jr., MD, MAEd, is Associate Professor of Nursing at the University of Alabama at Birmingham, School of Nursing.
Intended audience: This continuing education (CE) activity has been designed to meet the educational needs of nurse practitioners and other clinicians who provide women’s healthcare.
CE approval period: Now through June 30, 2024
Estimated time to complete this activity: 1 hour
CE approval hours: 1.0 contact hour of CE credit including 0.33 contact hours of pharmacology content
Goal statement: Nurse practitioners and other clinicians who provide women’s healthcare will increase their knowledge about evaluation and conservative management of urinary incontinence (UI).
Needs assessment: UI affects as many as 25% to 75% of women in the United States depending on age. Women are often not forthcoming about symptoms unless asked by their healthcare provider, suffering unnecessary reduction in quality of life. Knowledge about risk factors and the use of in-office assessment can improve timely evaluation and diagnosis. Treatment plans can be individualized to each patient’s needs and desires when healthcare providers are informed about conservative nonpharmacologic and pharmacologic management options.
Educational objectives: At the conclusion of this educational activity, participants should be able to:
- Identify elements of history, physical exam, and in-office testing for diagnosis of UI.
- Discuss recommended nonpharmacologic management for UI.
- Describe indications, mechanism of action, efficacy, adverse events, and contraindications for pharmacologic options in treating UI.
Accreditation statement: This activity has been evaluated and approved by the Continuing Education Approval Program of the National Association of Nurse Practitioners in Women’s Health (NPWH) and has been approved for 1 contact hour CE credit, including 0.33 hours of pharmacology credit.
Faculty disclosures: NPWH policy requires all faculty to disclose any affiliation or relationship with a commercial interest that may cause potential, real, or apparent conflict of interest with the content of a CE program. NPWH does not imply that affiliation or relationship will affect the content of the CE program. Disclosures provide participants with information that may be important to their evaluation of an activity.
Lisa S. Pair, APRN, DNP, WHNP-BC, has no actual or potential conflicts of interest in relation to the contents of this article.
William E. Somerall, Jr., MD, MAEd, has no actual or potential conflicts of interest in relation to the contents of this article.
Disclosure of unlabeled/unapproved use: NPWH policy requires authors to disclose to participants when they are presenting information about an unlabeled use of a commercial product or device or an investigational use of a drug or device not yet approved for any use.
Disclaimer: Participating faculty members determine the editorial content of the CE activity; this content does not necessarily represent the views of NPWH. This content has undergone a blinded peer review process for validation of clinical content. Although every effort has been made to ensure that the information is accurate, clinicians are responsible for evaluating this information in relation to generally accepted standards in their own communities and integrating the information in this activity with that of established recommendations of other authorities, national guidelines, FDA-approved package inserts, and individual patient characteristics.
Successful completion of the activity: Successful completion of this activity, J-22-03, requires participants to do the following:
- “Sign in” at the top right-hand corner of the page pathlms.com/npwh/courses/42979. If you have an NPWH account. You must be signed in to receive credit for this course. If you do not remember your username or password, please follow the “Forgot Password” link and instructions on the sign-in page. If you do not have an account, please click on “Create an Account.”*
- Read the learning objectives, disclosures, and disclaimers on the next page and then click on the “Continue” button.
- Study the material in the learning activity during the approval period (now through June 30, 2024).
- Complete the post-test and evaluation. You must earn a score of 70% or higher on the post-test to receive CE credit.
- Print out the CE certificate after you have successfully passed the post-test and completed the evaluation.
*If you are an NPWH member, were once a member, or have completed CE activities with NPWH in the past, you have a username and password in our system. Please do not create a new account. Creation of multiple accounts could result in loss of CE credits as well as other NPWH services. If you do not remember your username or password, either click on the “Forgot Username” or “Forgot Password” link or call the NPWH office at (202) 543-9693, ext. 1.
Commercial support: This activity did not receive any commercial support.
Urinary incontinence (UI) is underreported and undertreated in many women. Screening and evaluation are the first steps toward addressing this health issue. Optimizing continence through conservative management can improve the quality of life for women and decrease costs for individuals and the healthcare system. Primary care nurse practitioners can screen, evaluate, and initiate and monitor conservative management for UI based on patient preference with referral to a urogynecologist as needed.
Womens Healthcare. 2022;10(3):10-17. doi: 10.51256/WHC062210
Urinary incontinence (UI) is a common condition affecting as many as 25% to 75% of women depending on age that includes urge urinary incontinence (UUI), stress urinary incontinence (SUI), mixed urinary incontinence (MUI), and overactive bladder (OAB), with an estimated annual direct cost of $19.5 billion for incontinence care.1 UUI is considered a symptom of overactive bladder syndrome. Overactive bladder is characterized as urinary urgency, frequency, and nocturia with or without UUI in the absence of pathology.2 In the United States, it is estimated that as many as 36.4% of the population experience UUI.2 In addition, approximately 37.5% of women ages 30 to 50 years describe SUI during their primary care visits.3 This article describes the clinical symptoms, evaluation, and cost-effective nonsurgical, conservative management of urinary incontinence.
Etiology and risk factors for UI
Urinary incontinence is the “complaint of involuntary loss of urine.”4 Stress urinary incontinence is defined as “involuntary loss of urine on effort or physical exertion (eg, athletic activities) or on sneezing or coughing.”4 This occurs from urethral sphincter weakness or urethral hypermobility. Continence is maintained when the urethral closure pressure is greater than intraabdominal/bladder pressure. When intraabdominal/bladder pressure becomes greater than the urethral closure pressure, incontinence occurs. Urge urinary incontinence is “the involuntary loss of urine associated with urgency.”4 This occurs when the detrusor muscle of the bladder becomes overactive and contracts causing UI associated with urgency and sometimes with urinary frequency or nocturia. The cause of UUI may be idiopathic or a result of neurologic conditions such as multiple sclerosis, Parkinson disease, or diabetes mellitus type 2. Mixed urinary incontinence occurs when the individual experiences both SUI and UUI. Overflow incontinence results from incomplete bladder emptying due to inability of the detrusor muscle to contract optimally or because of a bladder outlet obstruction, so the bladder becomes too full and leaks. This can be caused by neurologic conditions, medication side effects, or pelvic organ prolapse. Individuals may experience similar symptoms of SUI and UUI because of incomplete bladder emptying. Functional incontinence results from the inability to physically reach the toilet in time because of mobility or cognitive impairments.4
Risk factors for UI include obesity, frequent heavy lifting, chronic coughing, advancing age, chronic medical conditions, certain medications such as diuretics, smoking, caffeine intake, genitourinary syndrome of menopause (GSM), sedentary behavior, hysterectomy, pregnancy, and childbirth. Working with individuals to reduce modifiable risk factors may improve continence.5–10
Screening and history
Screening patients for UI is an important first step as most individuals do not report symptoms of UI unless asked. The Women’s Preventive Services Initiative recommends screening women annually for UI and, if present, assessing the effect UI has on their quality of life. When UI is identified, asking questions to differentiate the type and determine the severity of distress of UI is critical to direct the evaluation and management plan. It is important to identify the onset, character, degree of bother to the individual, and treatment preference.10–13 A thorough history that reviews the individual’s medical, surgical, obstetric, gynecologic, and sexual history will help the provider narrow the differential diagnoses and reduce unnecessary laboratory or diagnostic tests.13 A medication review will help identify if medications are playing a part in the symptoms. Certain medications such as loop diuretics, adrenergic blockers, angiotensin-converting enzyme inhibitors (side effect, cough), sodium-glucose cotransporter-2 inhibitors, and muscle relaxants exhibit effects that can influence bladder function.10,14
Physical examination and other in-office assessment
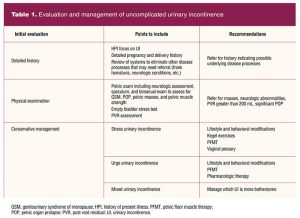
The physical examination should include a focused genitourinary system assessment. As the individual prepares for the exam, they are asked to empty their bladder and collect a clean-catch urine specimen. A point-of-care dipstick urinalysis is performed to rule out urinary tract infection (UTI), presence of hematuria, proteinuria, and glycosuria, all of which may indicate medical conditions needing referral. If suggestive for UTI, the urine is sent for culture and sensitivity and the individual is empirically treated for uncomplicated cystitis. Further assessment and management for UI is postponed until after UTI treatment (Table 1).1,10
The external genitalia are assessed for vulvar contact dermatitis exhibiting signs of excoriation, erythema, and wetness that results in a decrease in skin barrier function from overexposure to urine. Additionally, a neurologic evaluation of the S2–S4 nerve roots is performed by assessing for the anal reflex or the bulbocavernosus reflex. If neurologically intact, there is contraction of the external anal sphincter when stroking the skin adjacent to the anus bilaterally with a cotton tip applicator (anal reflex) or gently squeezing or tapping the clitoris (bulbocavernosus reflex).15 The vagina is assessed for pelvic floor prolapse, discharge, and pallor or loss of rugae as indicators of loss of estrogenic effect. Pelvic floor prolapse can affect bladder storage and emptying function. Anterior wall prolapse often results in difficulty of bladder emptying or UUI. Lack of urethral support can result in SUI. Vaginal discharge can be mistaken for UI, and individuals with GSM can experience UI symptoms.14
The bimanual exam will provide information on uterine size, presence of uterine or adnexal masses, and any tenderness that may indicate the need for further diagnostic evaluation. Pelvic floor muscle strength is assessed by placing the index and middle fingers into the vagina and asking the individual to squeeze the pelvic floor muscles. Some comments to share that help them identify and contract their pelvic floor muscles optimally would be to instruct the individual to squeeze the muscles as if they are trying to inhibit the accidental passage of flatus. Another way to help them isolate the pelvic muscles would be to tell them to contract the muscles that would be tightened when pants are pulled over the hips. The importance of identifying the individual’s ability to isolate and contract their pelvic floor muscles is helpful in determining treatment recommendations.16
An empty bladder stress test can be conducted as part of the evaluation. The individual is asked to bear down and cough while the provider observes for leaking. Positive leakage is suggestive of SUI. A post void residual (PVR) assessment is helpful to evaluate the bladder emptying function and to rule out overflow incontinence. This can be performed via straight catheterization or bladder ultrasound. Most primary care and women’s health clinics do not have a bladder ultrasound, so the test is performed with catheterization. The PVR assessment should occur within 10 minutes of emptying the bladder for an accurate assessment. There is no consensus on normal PVR, but the American College of Obstetricians and Gynecologists defines a PVR of less than 150 mL as normal. If the PVR is over 200 mL, the individual should be referred to a urogynecology specialist for further assessment.1,14
When history, physical exam, and in-office assessment tests indicate UI, imaging studies and urodynamic testing are not indicated for individuals who desire conservative treatment.1,16
Nonpharmacologic management of UI
The treatment for UI depends on the cause. First-line treatment recommendations for all types of UI include lifestyle modifications and behavioral training.14 A 3-day bladder diary may be helpful in analyzing voiding patterns, leakage episodes and associated symptoms, timing of voids, use of incontinence pads, and the type and volume of fluid intake. Review of the bladder diary results can be instrumental in directing management. An example of a bladder diary can be found at niddk.nih.gov/-/media/Files/Urologic-Diseases/diary_508.pdf.
Lifestyle modifications include limiting fluid intake to 6 to 8 glasses (8 ounces each) per day, reducing carbonated beverages and alcohol, and limiting caffeine intake to no more than 2 cups of coffee or tea per day.12,14 Although reducing caffeine intake does not improve UI, it improves symptoms of urgency and frequency.17 Behavioral therapy includes bladder training and pelvic floor exercises. Bladder training (also known as bladder drills or bladder re-education) is used primarily for OAB and UUI and includes patient education and strategies with the goal of extending voiding intervals, expanding bladder capacity, and decreasing incontinence episodes. This is accomplished through focusing on increased time between voids.10 A baseline bladder diary will help determine the voiding frequency. Those who void every hour would be instructed to void every hour and 15 minutes, even if they do not need to void. After several days of delaying voiding with no accidents, the time interval is increased by 15 minutes. The goal is to void every 2 to 3 hours without accidents while awake. For those experiencing the urgency to void before the scheduled voiding time, pelvic floor muscle exercises and distraction techniques are employed to suppress the urgency. Distraction from the urgency sensation to void can be achieved through mental diversion by performing mental math, making a to-do list, or other activities that demand focus.16,18 Behavioral therapy has been found to be more effective than no treatment or placebo.18 Furthermore, an 8% reduction in weight for individuals who are overweight or obese has been associated with improved continence.19 The PRIDE [Program to Reduce Incontinence by Diet and Exercise] randomized control trial found after 6 months, the weight loss group reported a reduction of at least 70% in the total weekly number of any incontinence episodes, stress incontinence, and urge incontinence, with SUI more affected than UUI.19
Conservative options for management of SUI include pelvic floor muscle exercises (ie, Kegel exercises) and the use of a vaginal pessary.9 With Kegel exercises, the individual is taught to optimally identify their pelvic floor muscles and strengthen the vaginal muscles without contracting the anus through focused contractions. These exercises can stabilize the urethra and increase urethral closure pressure. Instructions include squeezing the pelvic floor muscles tight and holding for 5 to 10 seconds, then relaxing for 10 seconds before squeezing again. This should be done 10 times per set, with three sets per day, morning, noon, and night. This exercise regimen should be maintained for at least 3 months to achieve optimal results.13 The individual can also contract their pelvic floor muscles when they perform activities that increase intraabdominal pressure. This will help prevent leakage. Patients unable to contract their pelvic floor muscles appropriately should be referred to a physical therapist or nurse practitioner who specializes in pelvic floor muscle therapy (PFMT).
The International Urogynecological Associates provides links to patient education on pelvic floor exercises via a handout and videos. These resources can be found at yourpelvicfloor.org/conditions/pelvic-floor-exercises/.
The incontinence pessary is a silicone device that is placed in the vagina that provides a backstop of support for the urethra and increases urethral resistance.1,16,20 The incontinence pessary is shaped as a ring or dish with a knob that comes in various sizes. Patients are fitted with the appropriate size that rests comfortably between the vaginal apex and behind the pubic arch. The patient is instructed to wear the pessary as needed, either all day, only during exercise, and then removing at night if desired.16 Alternatives to the pessary are several over-the-counter products. Devices include reusable flexible silicone intravaginal devices and a disposable single-use bladder support device available in three sizes. These devices function similar to a pessary and are inserted into the vagina using a tampon-like applicator.21,22 There are limited data, however, on the efficacy of these alternative methods.
Various types of absorbent incontinence products are available. The single-use disposable products include pads, pull-on pads, and all-in-ones (formerly adult briefs or diapers). Reusable washable products include cotton and nylon absorbent underwear (panties, briefs), pads, and liners.23,24
Practice guidelines do not provide timelines for follow-up. However, most providers recommended follow-up at 3 months to assess treatment response or to consider next steps.13 Those fitted with a pessary may benefit from a 6-week follow-up appointment. Instructing the patient to wear their pessary to the follow-up visit is important for assessing whether the pessary is placed correctly and comfortably. In addition, the visit may assist with identifying any device issues that can be corrected such as a refitting or education to prevent pessary use discontinuation. For those who are pursuing lifestyle and behavioral therapy, it may be beneficial if they return with a bladder diary.10 The patient can complete a 3-day diary before they begin treatment and at 3 months prior to their appointment. Assessing for success and barriers to lifestyle and behavioral therapy along with a review of the bladder diary can assist the provider in the use of motivational techniques and education.
Pharmacologic therapy for UI
Lifestyle modifications, behavioral training, Kegel exercises, PFMT, and continence pessaries remain the primary therapeutic options for SUI because there is no available US Food and Drug Administration (FDA)-approved pharmacologic management for this condition. For individuals with GSM and UI, however, the addition of hormonal support may offer significant benefit. This can be done with estrogen and with nonestrogen agents such as the selective estrogen receptor modulator ospemifene. Studies have shown that topical estrogen tends to reduce vulvovaginal atrophy and UI symptoms while systemic estrogen may increase UI symptoms.25 After 12 weeks, the use of ospemifene decreased the number of voiding episodes in 24 hours, reduced nocturia, and decreased the number of UI incidents.26 For individuals with UUI who have experienced only minimal-to-moderate success with nonpharmacologic therapeutic options, the addition of FDA-approved medications often provides significant symptomatic improvement.18,27
Understanding the underlying physiology of the sympathetic nervous system (SNS) and parasympathetic nervous system (PNS) in the storage of urine and bladder emptying provides the foundation for pharmacologic therapeutic intervention for OAB and UUI. Urinary continence and urine storage is controlled by the SNS with the coordinated relaxation of the detrusor muscle and contraction of the internal urethral sphincter. When the internal urethral sphincter pressure is greater than the intravesical pressure, no leaking occurs. Emptying the bladder is achieved by the coordinated PNS stimulation of the detrusor muscle causing bladder contraction with the simultaneous relaxation of the internal urethral sphincter. When intravesical pressure exceeds urethral pressure, bladder emptying occurs.28
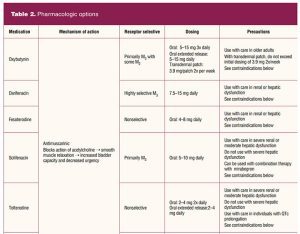
Pharmacologic treatment options include sympathetic beta-3 agonists that stimulate detrusor muscle relaxation and increase sphincter tone and muscarinic antagonists that block cholinergic receptor stimulation causing bladder relaxation (Table 2).
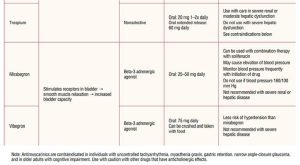
Because of receptor selectivity, the beta-3 agonists mirabegron and vibegron are the first-line drugs of choice. Although these drugs are more expensive than the older anticholinergic medications, receptor selectivity provides a reduced side-effect profile with greater chance of adherence to treatment. Mirabegron is available in an extended formulation generally reaching full therapeutic effect in 6 to 8 weeks. A side effect of mirabegron is hypertension. Careful blood pressure monitoring is required early in treatment, particularly in individuals with elevated blood pressure. Mirabegron should not be used in individuals with blood pressure 160/100 mm Hg or greater until appropriate blood pressure management is in place.29 Mirabegron can be combined with antimuscarinic medications if needed for improved symptom control.20 No dosing adjustment is needed for individuals with mild renal or hepatic dyfunction. For moderate renal or hepatic dysfunction, maximum daily dosing should not exceed initial daily dosing. Mirabegron is not recommended in severe renal or hepatic disease.29
Vibegron is not associated with hypertension and can be crushed and taken with food. Vibegron is also useful in individuals with mild-to-moderate liver impairment and in severe renal disease. It should not be used with severe liver impairment or if the eGFR is less than 15 mL/min/1.73 m2.30 It can be combined with antimuscarinic medications if needed for improved symptom control. With both mirabegron and vibegron, monitoring for urinary retention is needed.
Even though the antimuscarinic drugs have been available for many years, there has been little receptor specificity until relatively recently. Muscarinic receptors are found in many locations in the body. The muscarinic receptor subtypes are: M1 found in the central nervous system and the salivary glands; M2 receptors found in the heart; M3 receptors found in salivary glands, the detrusor muscle of the bladder, gastrointestinal tract smooth muscle, muscles in the eye (iris and the ciliary muscle), and the lacrimal glands.28 Because of the lack of receptor specificity for most anticholinergic drugs, side effects are common. The significant side effects are dry mouth, dry eyes, constipation, blurred vision, drowsiness, tachycardia, and an increased risk of dementia. Antimuscarinics are contraindicated in individuals with uncontrolled tachyarrhythmia, myasthenia gravis, gastric retention, narrow angle-closure glaucoma, and in older individuals with cognitive impairment.31 Available antimuscarinic medications include oxybutynin, trospium, darifenacin, fesoterodine, solifenacin, and tolterodine (Table 2).
As with most therapeutic interventions, it is important to begin with the lowest dose. Dosage can be reevaluated in 2 to 4 weeks and slowly increased if needed until therapeutic goals are met. If the individual remains symptomatic and cannot tolerate a dosage increase for either class of drugs, combination therapy with daily low-dose mirabegron 25 mg and solifenacin 5 mg is recommended. If response remains inadequate after 4 to 8 weeks, the mirabegron can be increased to 50 mg daily.30,32 Although data are limited, dual antimuscarinic therapy may be useful for individuals with partial antimuscarinic response who cannot take beta-3 agonists. Individuals using dual antimuscarinic therapy must be counseled on the risks and monitored for cognitive changes.30,32 There are limited human data in pregnancy for use of beta-3 agonists and antimuscarinic drugs. Also based on limited human use information, the beta-3 agonists and antimuscarinic drugs, except for tolterodine with potential toxicity, are considered “probably compatible” for use with breastfeeding.33
Implications for practice
Individuals are not always forthcoming about their symptoms of UI. Primary care providers should proactively screen all patients. When assessment indicates a diagnosis of uncomplicated UI, conservative management can be initiated with follow-up. Individuals not satisfied with conservative therapy; in whom diagnosis of UI is unclear; who experience associated pain, persistent hematuria, proteinuria, elevated PVR, or significant POP; have had previous pelvic surgery or radiation therapy; or who desire surgical intervention should be referred to a urogynecologist.1,13 Optimizing continence can improve quality of life while also decreasing the costs of incontinence care.
References:
- American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 155: urinary incontinence in women. Obstet Gynecol. 2015;126(5):e66-e81.
- Milsom I, Coyne KS, Nicholson S, et al. Global prevalence and economic burden of urgency urinary incontinence: a systematic review. Eur Urol. 2014;65(1)79-95.
- Ng SF, Lok MK, Pang SM, Wun YT. Stress urinary incontinence in younger women in primary care: prevalence and opportunistic intervention. J Womens Health (Larchmt). 2014;23(1):65-68.
- Haylen BT, de Ridder D, Freeman RM, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic floor dysfunction. Int Urogynecol J. 2010;21(1):5-26.
- Juliato CR, Baccaro LF, Pedro AO, et al. Factors associated with urinary incontinence in middle-aged women: a population-based household survey. Int Urogynecol J. 2017;28(3):423-429.
- Longo PS, Borbily LV, Glina FPA. Urinary incontinence following subtotal and total hysterectomy: a systematic review. Einstein (Sao Paulo). 2019;17(2):eRW4320.
- Dicpinigaitis PV. Prevalence of stress urinary incontinence in women presenting for evaluation of chronic cough. ERJ Open Res. 2021;7(1):00012-2021.
- Le Berre M, Presse N, Morin M, et al. What do we really know about the role of caffeine on urinary tract symptoms? A scoping review on caffeine consumption and lower urinary tract symptoms in adults. Neurourol Urodyn. 2020;39(5):1217-1233.
- Wang K, Xu X, Jia G, Jiang H. Risk factors for postpartum stress urinary incontinence: a systematic review and meta-analysis. Reprod Sci. 2020;27(12):2129-2145.
- Gormley EA, Lightner DJ, Burgio KL, et al. Diagnosis and treatment of overactive bladder (non-neurogenic) in adults: AUA/SUFU guideline. J Urol. 2012;188(6 suppl):2455-2463.
- Women’s Preventive Services Initiative. Screening for urinary incontinence. 2018.
- American Urogynecologic Society and American College of Obstetricians and Gynecologists. Committee opinion: evaluation of uncomplicated stress urinary incontinence in women before surgical treatment. Female Pelvic Med Reconstr Surg. 2014;20(5):248-251.
- Sussman RD, Syan R, Brucker BM. Guideline of guidelines: urinary incontinence in women. BJU Int. 2020;125(5):638-655.
- Abrams P, Andersson KE, Apostolidis A, et al. 6th International Consultation on Incontinence. Recommendations of the International Scientific Committee: evaluation and treatment of urinary incontinence, pelvic organ prolapse and faecal incontinence. Neurourol Urodyn. 2018;37(7):2271-2272.
- Hull MA. Assessment of women. In: Newman DK, Whyman JF, Welch VW,eds. SUNA Core Curriculum for Urologic Nursing, 1st ed. Anthony J Janetti, Inc; 2017:235-246.
- Handler SJ, Rosenman AE. Urinary incontinence: evaluation and management. Clin Obstet Gynecol. 2019;62(4):700-711.
- Hannestad YS, Rortveit G, Daltveit AK, Hunskaar S. Are smoking and other lifestyle factors associated with female urinary incontinence? The Norwegian EPINCONT Study. BJOG. 2003;110(3):247-254.
- Balk E, Adam GP, Kimmel H, et al. Nonsurgical treatments for urinary incontinence in women: a systematic review update. Agency for Healthcare Research and Quality; 2018.
- Subak LL, Wing R, West DS, et al; PRIDE Investigators. Weight loss to treat urinary incontinence in overweight and obese women. N Engl J Med. 2009;360(5):481-490.
- Lipp A, Shaw C, Glavind K. Mechanical devices for urinary incontinence in women. Cochrane Database Syst Rev. 2014;2014(12):CD001756.
- Ziv E, Stanton SL, Abarbanel J. Efficacy and safety of a novel disposable intravaginal device for treating stress urinary incontinence. Am J Obstet Gynecol. 2008;198(5):594.e1-594.e5947.
- Simpson AN, Garbens A, Dossa F, et al. A cost-utility analysis of nonsurgical treatments for stress urinary incontinence in women. Female Pelvic Med Reconstr Surg. 2019;25(1):49-55.
- National Association for Continence. Adult absorbent products.
- Fader M, Cottenden A, Chatterton C, et al. An International Continence Society (ICS) report on the terminology for single-use body worn absorbent incontinence products. Neurourol Urodyn. 2020;39(8):2031-2039.
- Bodner-Adler B, Alarab M, Ruiz-Zapata AM, Latthe P. Effectiveness of hormones in postmenopausal pelvic floor dysfunction. International Urogynecological Association research and development-committee opinion. Int Urogynecol J. 2020;31(8):1577-1582.
- Schiavi MC, Zullo MA, Faiano P, et al. Retrospective analysis in 46 women with vulvovaginal atrophy treated with ospemifene for 12 weeks: improvement in overactive bladder symptoms. Gynecol Endocrinol. 2017;33(12):942-945.
- Burgio KL, Locher JL, Goode PS. Combined behavioral and drug therapy for urge incontinence in older women. J Am Geriatr Soc. 2000;48(4):370-374.
- Drugs for overactive bladder. In: Burchum JR, Rosenthal LD, eds. Lehne’s Pharmacology for Nursing Care, 11th ed. Philadelphia, PA: Elsevier; 2022: 134-139.
- Mybetriq [mirabegron]. US Food and Drug Administration approved product information. Revised April 2021.
- Gemtesa [vibegron]. US Food and Drug Administration approved product information. Revised December 2020.
- Gratzke C, Chapple C, Mueller ER, et al. Efficacy and safety of combination pharmacotherapy for patients with overactive bladder: a rapid evidence assessment. Eur Urol. 2019;76(6):767-779.
- Diamond SE. Overactive bladder. In: Arcangelo VP, Peterson AM, Wilbur VF, Kang TM, eds. Pharmacotherapeutics for Advanced Practice, 5th ed. Philadelphia, PA: Wolters Kluwer; 2022: 595-613.
- Briggs GG, Towers CV, Forinash AB, eds. Drugs in Lactation and Pregnancy, 12th ed. Philadelphia, PA: Wolters Kluwer; 2022.
Key words: urinary incontinence, urinary incontinence evaluation, management of urinary incontinence, stress urinary incontinence, urge urinary incontinence, overactive bladder
Womens Healthcare. 2022;10(3):10-17. doi:10.51256/WHC062210