The first aim of this study was to identify characteristic differences between college women who completed the 3-dose human papillomavirus (HPV) vaccine series prior to first sexual intercourse and those who did not. The second aim was to identify the knowledge and social influences associated with successful HPV vaccine completion prior to the first intercourse. Results of this study, which have useful implications for healthcare providers caring for adolescents and college-age women, are presented.
Human papillomavirus (HPV) is the most common sexually transmitted infection in the United States.1 In fact, almost all sexually active women and men contract HPV at some point in their lives. Among more than 100 HPV strains, about 40 cause genital warts or cancer, including cervical, vulvar, vaginal, anal, and oropharyngeal cancers.1 HPV vaccination can protect females and males against most HPV types that cause genital warts or cancer.
National recommendations for HPV vaccination have changed over time. Since 2016, based on the latest immunogenicity data, the Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention has recommended routine HPV vaccination at age 11 or 12 years to ensure that the vaccine is effective before sexual initiation.2,3 (Vaccination can be started at age 9 years.) The ACIP also recommends vaccination for females aged 13 to 26 years and males aged 13 to 21 years not adequately vaccinated previously. Vaccination is recommended through age 26 years for gay, bisexual, and other men who have sex with men; transgender persons; and immunocompromised persons (including those with HIV infection) not adequately vaccinated previously.
Current HPV vaccination coverage rates among persons aged 19 to 26 years are 47% to 52% for women (full coverage) and 14% to 21% for men (at least one dose).4 Mean ages of first intercourse for females and males are 17.3 years and 17.0 years, respectively.5 The relatively low rate of HPV vaccination among persons likely to be engaging in sexual intercourse is a concern because vaccine completion is needed before sexual initiation to be most effective. Healthcare providers (HCPs) who care for adolescents and young adults need to understand the factors that influence HPV vaccine uptake so that they can help increase the rate of HPV vaccination in their practices.
Although vaccination behaviors are influenced by many factors, a critical evaluation of the social influences of parents, sexual partners, peers, and HCPs on young persons’ HPV vaccination behaviors would be elucidating.6–8 Thus far, studies have shown that these individuals/groups can have a favorable effect on vaccine uptake among young persons.9–18 A deeper investigation was needed to determine the influences of these individuals and groups on HPV vaccination before sexual initiation.
Purpose
This study expanded the current literature by focusing on college women and their HPV vaccination status prior to first sexual intercourse. In particular, this study was conducted to: identify characteristic differences between college women who completed the 3-dose HPV vaccine series prior to first sexual intercourse and those who did not complete the series before their first sexual intercourse; and identify the knowledge and social influences associated with HPV vaccine completion prior to first sexual intercourse. This study focused on females because they are disproportionately burdened by HPV-related cancers.19
Methods
Study population
A purposive sample of college women was recruited via email advertisements to participate in a study that entailed completion of an online questionnaire during spring 2011. (Of note, men were initially included in the study but were ultimately excluded because of an oversight/shortcoming in the original study design. Questions regarding HPV vaccination status were posed only to female participants.) The study was conducted at two large Texas universities over a 2-week period. Each university’s Institutional Review Board approved the study. Emails were distributed using each university’s regulated delivery system. The body of the email contained a link to the internet-based questionnaire. Two questionnaire reminder emails were sent 5 and 10 days after the initial recruitment email.
Participation in this study was voluntary, and no identifiable information was collected. Participants received no compensation. Inclusion criteria were as follows: self-identification as female, age ≤ 26 years, and enrollment in one of the two universities. Participants were excluded if they had a history of cervical cancer, had missing data regarding a family history of cervical cancer, and/or had never initiated the HPV vaccine series. In addition, participants who were vaccinated at the same age at which they first had their sexual intercourse were removed because of possible temporal bias.
Measures Dependent variables
Some questionnaire items were previously tested and validated.20,21 Others, informed by available literature, were developed and adapted by the researchers for the study. The timing of participants’ HPV vaccination status relative to the timing of their first sexual intercourse was determined by asking them two questions, the answers to which needed to be provided in years: “How old were you when you had sexual intercourse for the first time?” and “At what age did you receive your last HPV vaccine shot?” (In this context, “last” refers specifically to the last shot of the vaccination series.) The authors relied on these self-reported data. They were unable to verify vaccination through health records. If the age that participants provided for receiving their last HPV shot was younger than that provided for their first sexual intercourse, they were coded “yes,” and if they were older when they received their last HPV shot than when they had their first sexual intercourse, they were coded “no.”
Independent variables
Participants answered 11 true-false questions assessing their HPV knowledge.21 Correctly endorsed items were coded as “1,” and incorrectly endorsed items were coded as “0.” The number of correct items (0–11) was summed; higher scores reflected greater HPV-related knowledge. Participants were asked about their perceptions of cancer risk. In particular, they were asked if any of their immediate-family members had cervical cancer. Response options were “yes” or “no.” They also were asked the number of their friends who had already received the HPV vaccine.22 Response options were “none, some, most or all,” and “I don’t know.” With regard to the norm of vaccination, participants were asked, “How much did the opinion of the following people influence your decision about getting the HPV vaccine?” Response categories were scored on a 4-point Likert scale and ranged from “very much” to “not at all.” Participants responded to this item separately for each of these persons: “your parents, your best friend, your religious leader,” and “your healthcare provider.”
Statistical analyses
All statistical analyses were performed using SPSS (version 22). Frequencies were calculated for all major study variables. Distribution frequencies were then compared by participants’ HPV vaccination completion status. Univariate statistics were performed to identify differences for variables. A binary logistic regression model was fitted to identify factors associated with 3-dose HPV vaccine completion prior to first sexual intercourse as compared to those who completed vaccination after the first sexual intercourse.
Results
A total of 81,715 enrolled college students, both women and men, received the invitation email. Among this group, 3,987 initiated the survey (response rate, 4.9%) and 2,513 completed it (completion rate, 63.1%). After excluding participants for missing data (n = 136), reporting their sex as male (n = 543), being at least age 26 years (n = 469), or having a past history of cervical cancer (n = 16), as well as those reporting never initiating HPV vaccination (n = 846), the final sample size for analysis was 503 women.
Sample characteristics, HPV knowledge scores, and perceptions of cancer risk
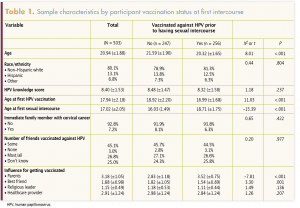
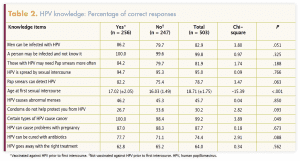
Table 1 provides sample characteristics of the women included in this analysis. Table 2 lists the HPV knowledge questions and the proportions of the “no” group, the “yes” group, and the entire group who answered correctly. Among all participants, 7.2% reported that an immediate family member had been diagnosed with cervical cancer. Again, among all participants, 45.1% reported that some of their friends were vaccinated and 25.0% said they did not know their friends’ vaccination status.
Multivariate analysis
Factors associated with HPV vaccine completion prior to first sexual intercourse are presented in Table 3. Participants who were younger (odds ratio [OR], 0.70; 95% confidence interval [CI], 0.61–0.79) and who reported being influenced by their parents (OR, 2.00; 95% CI, 1.59–2.51) or their best friends (OR, 0.67; 95% CI, 0.54–0.85) were more likely to be HPV vaccine complete prior to first sexual intercourse; the difference was statistically significant. Factors such as race/ethnicity, immediate family history of cervical cancer, friends vaccinated against HPV, and influence from religious leaders and HCPs were not statistically significantly associated with HPV vaccine completion prior to first sexual intercourse.
Discussion
In this study of college women in Texas, the average age of HPV vaccination was approximately age 18 years and only half were fully vaccinated prior to first sexual intercourse. Young women whose parents supported HPV vaccination had the greatest odds 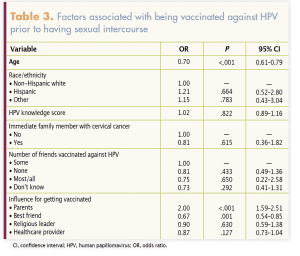 of obtaining the vaccine prior to first intercourse. A clear need exists for improved vaccine communication among HCPs, young female patients, and their parents as a strategy to increase HPV vaccination at recommended ages (11–12 years) to ensure optimal protection. Because of low on-time HPV vaccination in so many college students, HCPs caring for members of this population should ensure that education about HPV and HPV vaccination is reinforced and that vaccine services are clearly promoted and delivered.
of obtaining the vaccine prior to first intercourse. A clear need exists for improved vaccine communication among HCPs, young female patients, and their parents as a strategy to increase HPV vaccination at recommended ages (11–12 years) to ensure optimal protection. Because of low on-time HPV vaccination in so many college students, HCPs caring for members of this population should ensure that education about HPV and HPV vaccination is reinforced and that vaccine services are clearly promoted and delivered.
Regarding college women’s HPV knowledge, the average score in this study was 8.4 out of 11 (76%), indicating that this sample was knowledgeable about HPV. Nevertheless, there was not much difference in HPV knowledge between women who were vaccinated before initiating sexual intercourse and those who were not, possibly suggesting that knowledge about HPV does not equate to behavior change—in this case, getting the full HPV vaccination series before sexual intercourse. These findings are aligned with those of a study evaluating an HPV vaccine completion intervention. Although HPV knowledge increased in a sample of college students, this knowledge did not translate to HPV vaccine completion.23 College health interventions focused on increasing HPV vaccine completion should extend beyond knowledge to include other known influences such as social normalization and vaccine-positive environments (including cues to vaccinate at health services and strong college HCP recommendations).
Parental influence on vaccination status found in this study is not unique.13,14,24 However, this study provides insight into parental influence prior to sexual intercourse initiation. Public health campaigns and HCPs need to ensure that parents understand the importance of HPV vaccination and the optimal timing of it. If vaccination has not occurred at the recommended age, parents can still encourage their teenaged and college-aged daughters to initiate and complete the series.13,14,24,25 HCPs can assist by disseminating information about the importance of the timing of HPV vaccination to ensure optimal cancer protection.22,26,27
In the present study, HCPs did not have a significant influence on college-age women’s HPV vaccination rates prior to intercourse. However, other studies have shown that strong HCP recommendations for HPV vaccination are a significant predictor of vaccine initiation.28 Some HCPs may provide weak recommendations or none at all.29 In addition, some may not adequately educate parents and patients about HPV.30,31 HCPs can help increase overall vaccination rates by providing strong recommendations, as well as education to enhance parents’ and patients’ HPV knowledge.32
Limitations
These data were cross-sectional and collected from students enrolled in two different colleges in Texas. They may not be generalized to all college-aged women, especially in other US regions. Inclusion and exclusion criteria limited the sample size, which further affects generalizability of the study. In addition, because items regarding vaccination status for male participants were lacking, men were excluded from the study, further limiting its generalizability. The survey relied on self-reported data, which could lead to a potential for bias in answering questions based on perceived social norms or to respondents providing incorrect information about their vaccine initiation/completion status or their age at sexual intercourse initiation. Despite these limitations, this study adds to the current literature and highlights the still-too-large proportion of young women who have not completed the HPV vaccination series by the recommended age.
Implications for practice
This study has produced three important findings: delayed vaccination does occur (among the sample, the mean age of HPV vaccine completion was nearly 18 years, which is 6 to 7 years beyond national vaccine age recommendations); only half of these women were vaccinated prior to their first sexual intercourse; and HPV vaccine-supportive parents were the strongest predictor of vaccination prior to first sexual intercourse. Although much is known about factors affecting parents’ and patients’ decision to initiate the HPV vaccination series, little is known about influences related to timing of vaccination and, in particular, timing prior to first sexual intercourse. This study provides insight into factors—age and influence of parents and best friends—associated with HPV vaccination.
Future research and quality improvement projects should focus on pediatric practices. However, the results of this study indicate that a focused effort is needed in college health practices as well. In particular, women’s health nurse practitioners and other advanced practice registered nurses can take the lead in promoting HPV vaccination on college campuses. College students aged 18 years or older can make their own healthcare decisions. Therefore, consistent HPV messaging on college campuses is needed, and clear recommendations from HCPs caring for college students are imperative.33 University level and provider level assessments of health services and health services training may be necessary, particularly at universities in low vaccine uptake regions such as Texas to decrease regional HPV vaccine disparities.
Additional research must include the HPV vaccination status of college men, an underrepresented population in the literature on HPV. Attention should be focused on HPV vaccination in males not only because it helps protect women against HPV transmitted to them by males but also because it protects males themselves. HPV infection can cause genital warts and penile, anal, and oropharyngeal cancers in men.3 Future interventions must expand beyond educational efforts. Interventions also need to improve the skills of HCPs in delivering strong vaccine recommendations, addressing parental or patient hesitancy, and creating HPV vaccine-positive environments, with opportunities to vaccinate, especially on college campuses.
Brittany L. Rosen is Assistant Professor at the School of Human Services, University of Cincinnati, in Cincinnati, Ohio. Leigh E. Szucs is Research Affiliate and Kelly L. Wilson is Associate Professor, both in the Department of Health and Kinesiology at Texas A&M University in College Station. Jovanni V. Reyes is a Research Fellow in the Oak Ridge Associated Universities in Atlanta, Georgia. Holly B. Fontenot is Associate Professor at Connell School of Nursing, Boston College, in Chestnut Hill, Massachusetts. Matthew Lee Smith is Co-Director of the Texas A&M Center for Population Health and Aging and Associate Professor at the Department of Environmental and Occupational Health, School of Public Health, at Texas A&M University in College Station. The authors state that they do not have a financial interest in or other relationship with any commercial product named in this article.
References
- CDC. Human papillomavirus (HPV): what is HPV? Updated December 13, 2016. cdc.gov/hpv/parents/whatishpv.html.
- McNair KT, Fontenot HB. Understanding the two-dose HPV vaccine schedule. Nurs Womens Health. 2018;22(2):175-180.
- CDC. HPV vaccine recommendations. Updated December 15, 2016. cdc.gov/vaccines/vpd/hpv/hcp/recommendations.html.
- CDC. Vaccination coverage among adults in the United States, National Health Interview Survey, 2016. Updated February 26, 2018. cdc.gov/vaccines/imz-managers/coverage/adultvaxview/pubs-resources/NHIS-2016.html#hpv.
- CDC. Key statistics from the National Survey of Family Growth – S listing. Updated August 14, 2017. cdc.gov/nchs/nsfg/key_statistics/s.htm#vaginalsexual.
- Boehner CW, Howe SR, Bernstein DI, Rosenthal SL. Viral sexually transmitted disease vaccine acceptability among college students. Sex Transm Dis. 2003;30(10):774-778.
- Brunson EK. The impact of social networks on parents’ vaccination decision. Pediatrics. 2013;131(5):e1397-e1404.
- Edberg M. Essentials of Health Behavior Social and Behavioral Theory in Public Health. 2nd ed. Burlington, MA: Jones & Bartlett; 2015.
- Allen JD, Mohllajee AP, Shelton RC, et al. Stage of adoption of the human papillomavirus vaccine among college women. Prev Med. 2009;48(5):420-425.
- Ratanasiripong NT, Cheng A, Enriquez M. What college women know, think, and do about human papillomavirus (HPV) and HPV vaccine. Vaccine. 2013;31(10):1370-1376.
- Gainforth HL, Cao W, Latimer- Cheung AE. Determinants of human papillomavirus (HPV) vaccination intent among three Canadian target groups. J Cancer Educ. 2012;27(4):717-724.
- Sandfort JR, Pleasant A. Knowledge, attitudes, and informational behaviors of college students in regards to the human papillomavirus. J Am Coll Health. 2009;58(2):141-149.
- Roberts ME, Gerrard M, Reimer R, Gibbons FX. Mother-daughter communication and human papillomavirus vaccine uptake by college students. Pediatrics. 2010;125(5):982-989.
- Rosenthal SL, Weiss TW, Zimet GD, et al. Predictors of HPV vaccine uptake among women aged 19-26: importance of a physician’s recommendation. Vaccine. 2011;29(5):890-895.
- Dorell C, Yankey D, Jeyarajah J, et al. Delay and refusal of human papillomavirus vaccine for girls, national immunization Survey–Teen, 2010. Clin Pediatr. 2014;53(3):261-269.
- Finney Rutten LJ, St. Sauver JL, Beebe TJ, et al. Association of both consistency and strength of self-reported clinician recommendation for HPV vaccination and HPV vaccine uptake among 11- to 12-year-old children. Vaccine. 2017;35(45):6122-6128.
- Ylitalo KR, Lee H, Mehta NK. Health care provider recommendation, human papillomavirus vaccination, and race/ethnicity in the US national immunization survey. Am J Public Health. 2013;103(1):164-169.
- Rahman M, Laz TH, McGrath CJ, Berenson AB. Provider recommendation mediates the relationship between parental human papillomavirus (HPV) vaccine awareness and HPV vaccine initiation and completion among 13- to 17-year-old U.S. adolescent children. Clin Pediatr (Phila). 2015;54(4):371-375.
- CDC. Human papillomavirus (HPV) and cancer. Updated August 29, 2018. cdc.gov/cancer/hpv/index.htm.
- CDC. Behavioral Risk Factor Surveillance System 2010 Questionnaire. Updated November 18, 2009. cdc.gov/brfss/annual_data/pdf-ques/2010brfss.pdf.
- Kahn JA, Rosenthal SL, Hamann T, Bernstein DI. Attitudes about human papillomavirus vaccine in young women. Int J STD AIDS. 2003;14(5):300-306.
- Geshnizjani A, Jozkowski KN, Middlestadt SE. Factors influencing the intention of getting the HPV vaccine among college women: an application of the reasoned action approach. Calif J Health Promot. 2013;11(2):1-11.
- Richman AR, Maddy L, Torres E, Goldberg EJ. A randomized intervention study to evaluate whether electronic messaging can increase human papillomavirus vaccine completion and knowledge among college students. J Am Coll Health. 2016;64(4):269-278.
- Dempsey AF, Zimet GD, Davis RL, Koutsky L. Factors that are associated with parental acceptance of human papillomavirus vaccines: a randomized intervention study of written information about HPV. Pediatrics. 2006;117(5):1486-1493.
- Wilson KL, Cowart CJ, Rosen BL, et al. Characteristics associated with HPV diagnosis and perceived risk for cervical cancer among unmarried, sexually active college women. J Cancer Educ. 2018;33(2):404-416.
- Caskey R, Lindau ST, Alexander GC. Knowledge and early adoption of the HPV vaccine among girls and young women: results of a national survey. J Adolesc Health. 2009;45(5):453-462.
- Ogilvie G, Anderson M, Marra F, et al. A population-based evaluation of a publicly funded, school-based HPV vaccine program in British Columbia, Canada: parental factors associated with HPV vaccine receipt. PLoS Med. 2010;7(5):e1000270.
- Holman DM, Benard V, Roland KB, et al. Barriers to human papillomavirus vaccination among US adolescents: a systematic review of the literature. JAMA Pediatr. 2014;168(1):76-82.
- Gilkey MB, Malo TL, Shah PD, et al. Quality of physician communication about human papillomavirus vaccine: findings from a national survey. Cancer Epidemiol Biomarkers Prev. 2015;24(11):1673-1679.
- Serpell L, Green J. Parental decision-making in childhood vaccination. Vaccine. 2006;24(19):4041-4046.
- Nagaraj A. Does qualitative synthesis of anecdotal evidence with that from scientific research help in understanding public health issues: a review of low MMR uptake. Eur J Public Health. 2006;16(1):85-88.
- Schnatz PF, Humphrey K, O’Sullivan DM. Assessment of the perceptions and administration of the human papillomavirus vaccine. J Low Genit Tract Dis. 2010;14(2):103-107.
- Jones M, Cook R. Intent to receive an HPV vaccine among university men and women and implications for vaccine administration. J Am Coll Health. 2008;57(1):23-31.

