Continuing education approval: This activity has been evaluated and approved by the Continuing Education Approval Program of the National Association of Nurse Practitioners in Women’s Health (NPWH) for 1.0 continuing education contact hours including 0.25 contact hours of pharmacology content now through August 31, 2025.
Educational objectives: At the conclusion of this educational activity, participants should be able to:
1. Describe cardiovascular, metabolic, and bone health implications for women with early loss of estrogen.
2. Discuss the implications of early estrogen loss on cognitive function, mood, sleep, and sexual health.
3. Discuss the use of hormone replacement therapy to decrease long-term health risks and bothersome symptoms associated with early estrogen loss.
The author has no actual or potential conflicts of interest in relation to the contents of this article.
Disclaimer: Participating faculty members determine the editorial content of the CE activity; this content does not necessarily represent the views of NPWH. This content has undergone a blinded peer review process for validation of clinical content. Although every effort has been made to ensure that the information is accurate, clinicians are responsible for evaluating this information in relation to generally accepted standards of care and integrating the information in this activity with that of established recommendations of other authorities, national guidelines, and individual patient characteristics.
Commercial support: This activity did not receive any commercial support.
Successful completion of the activity: Successful completion of this activity, J-23-04, requires participants to do the following:
1. “Sign in” at the top right-hand corner of the page pathlms.com/npwh/courses/54616 if you have an NPWH account. You must be signed in to receive credit for this course. If you do not remember your username or password, please follow the “Forgot Password” link and instructions on the sign-in page. If you do not have an account, please click on the “Create an Account.”*
2. Read the learning objectives, disclosures, and disclaimers.
3. Study the material in the learning activity during the approval period (now through August 31, 2025).
4. Complete the post-test and evaluation. You must earn a score of 70% or higher on the post-test to receive CE credit.
5. Print out the CE certificate after you have successfully passed the post-test and completed the evaluation.
*If you are an NPWH member, were once a member, or have completed CE activities with NPWH in the past, you have a username and password in our system. Please do not create a new account. Creation of multiple accounts could result in loss of CE credits as well as other NPWH services. If you do not remember your username or password, either click on the “Forgot Username” or “Forgot Password” link or call the NPWH office at (202) 543-9693, ext. 1.
Menopause is defined as the permanent cessation of menses caused by the loss of ovarian follicular activity. Physiologic or natural menopause occurs between the ages of 46 and 55 years, with a median age of 51 years. The diagnosis is confirmed retrospectively following 12 consecutive months of amenorrhea.1 Although most women experience menopause during this timeframe, approximately 5% of women will enter menopause earlier, between 40 and 45 years of age. An additional 1% of women will experience menopause prior to the age of 40 years, which is defined as premature menopause.2–4
Multiple etiologies have been associated with early loss of ovarian function. These include primary ovarian insufficiency, genetic disorders, infections, autoimmune disorders, or iatrogenic events due to surgery, chemotherapy, or pelvic radiation.1,3,4
Women with primary ovarian insufficiency (POI), or hypogonadotropic hypogonadism, experience loss of ovarian function prior to the age of 40 years. However, intermittent resumption of ovarian function can occur, resulting in spontaneous pregnancy for 5% to 10 % of women affected with this syndrome.1,5–7 Women with early loss of ovarian function commonly experience more significant hypoestrogenic symptoms. These include hot flashes, night sweats, sleep disruption, urogenital atrophy, and reduced quality of life.4,8 Symptoms can be gradual or abrupt in onset. Women undergoing surgical or chemotherapy-induced menopause experience more abrupt symptomatology.3,4
Irrespective of cause, women who experience early loss of estrogen are at increased risk for long-term health risks when compared to women who enter menopause at the average age.1 These include cardiovascular disease, diabetes, osteoporosis, dementia, depression, sexual dysfunction, and overall mortality.3,7–9 This article reviews the evidence on the association of early estrogen loss with long-term morbidities and the benefits of hormone therapy (HT) to mitigate risks and to alleviate hypoestrogenic symptoms. Management considerations are discussed.
Pathophysiology
The ovaries primarily secrete estrogen, progesterone, and testosterone during the premenopausal years. Although estrogen is responsible for the development and regulation of reproductive function in women, it also exerts a significant influence on other organs and body systems. This effect is mediated through signaling and activation of various estrogen receptors on target tissues that produce a tissue-specific response.2,10
Estrogen enhances endothelium-dependent vascular function and regulates anti-atherogenic activity in the cardiovascular system. Estrogen also suppresses neuroinflammation in the brain and promotes remodeling of synaptic connections. Additional beneficial effects include the regulation of mitochondrial metabolism and insulin sensitivity in muscles and the reduction of oxidative stress in various organs and tissues. The decrease in endogenous estrogen production associated with early hypoestrogenism leads to long-term health conditions including acceleration of cellular aging and quality of life.10
Observational data suggest that HT lowers the long-term morbidities associated with early estrogen loss.1–4,6,7,11–14 As such, the North American Menopause Society, the American Society for Reproductive Medicine, the American College of Obstetricians and Gynecologists, the International Menopause Society, and the European Society of Human Reproduction and Embryology have established clinical guidelines recommending HT for women with early estrogen loss until the expected age of natural menopause.1–9,11,13–17
Cardiovascular and metabolic implications
Cardiovascular disease (CVD) most often occurs in women following natural menopause or after age 55 years.1,6 The development of metabolic syndrome and progression of carotid atherosclerosis begins in the late perimenopause. Increases in low-density-lipoprotein (LDL) cholesterol and total cholesterol have been observed within the first 12 months following the final menstrual period (FMP). Various physiologic mechanisms have been associated with this hypoestrogenic milieu. These include impaired vasculature endothelial function, alterations in coagulation balance, diminished vascular flow, vasoconstriction, increased plasma levels of nitric oxide, and decreased levels of angiotensin-converting enzyme.1,4,8
Premature or early menopause is an established risk factor for CVD and death in women.1,3,4,18 This association has been supported by several observational studies including the Framingham study, which showed an increased CVD incidence in young postmenopausal women when compared to an age-matched control group. These results led to the initial hypothesis that estrogen is cardioprotective in women.3,4 Similarly, a recent meta-analysis of approximately 300,000 women noted an increased CVD risk in women with early menopause compared to women experiencing natural menopause at age 50 years or older. The greatest risk was identified in women with early bilateral salpingo-oophorectomy (BSO).3
Estrogen deprivation due to early BSO is associated with significant long-term adverse health outcomes.11,17 Data from several large observational studies have shown that although early BSO is associated with lower risks of breast and ovarian cancer, it is related to higher risks of all-cause mortality, coronary heart disease, parkinsonism, cognitive decline, osteoporosis, and multiple age-related morbidities.11 Data from the Mayo Clinic Cohort Study of Oophorectomy and Aging showed an increased risk of overall mortality and cardiovascular mortality in women with BSO before age 45 years. These risks were attenuated in a referent group of women who received HT. Similarly, the Nurse’s Health Study found that women with BSO younger than age 50 years had a significant increase in all-cause mortality and CVD mortality if they never used HT.3,4,17
Bone health
Bone remodeling in women is directly affected by circulating levels of estrogen. When bone resorption exceeds bone formation in estrogen-deficient states, a net bone loss occurs.4 Beginning 1 to 3 years prior to the FMP, an estimated 2.4% bone loss occurs annually over the next 5 to 10 years.3,4 Although rates of bone resorption subsequently decline, an approximate 30% reduction in peak bone mass is observed in women by 80 years of age.1
The use of HT for prevention of bone loss in women with early menopause has been supported by multiple clinical studies.4,6,8,13 A cross-sectional study comparing age and type of menopause (physiologic, early, and early BSO) on bone mineral density (BMD) found that women with early menopause had lower T scores of the vertebral spine and femoral neck when compared to those with natural menopause in the 45- to 55-year age group, with no statistical difference seen by ages 65 to 70 years. Similarly, a lower vertebral BMD was seen in women with early BSO compared to those with natural menopause between ages 45 and 50 years, with T scores remaining stable through age 65 to 70 years.4 In one prospective study, bone loss in women with early BSO was twofold higher when compared to women with natural menopause. Within 1 year, these women showed a significant increase in urinary N-telopeptide excretion and a significant decline in BMD when compared to their age-matched cohort.3,19
Cognitive function
Estrogen receptors are abundant in the hippocampus and prefrontal cortex of the brain. These areas regulate aspects of memory, learning, and higher-order cognitive function.1 Menopause stage and menopause symptoms appear to impact cognitive function at midlife.1,4 During the menopausal transition and early menopause, difficulties with concentration and recall have been reported. In the Penn Ovarian Age Study, a significant decline in verbal memory and processing speed was noted as women transitioned from the premenopause to postmenopause stage of the continuum.1,4 Similarly, menopausal symptoms such as anxiety, depression, and sleep disruption have been associated with cognitive difficulties. These include lower processing speed and poorer memory and attention.1,6
A decline in cognitive function is typically not associated with oophorectomy after natural menopause.4 However, findings from the Mayo Clinic Cohort Study of Oophorectomy and Aging found that early BSO was associated with a twofold increased risk of cognitive impairment or dementia. Similar findings were noted in another replicated study. Both studies identified a trend of higher risk that was correlated with earlier age at BSO. This risk was attenuated with use of HT at least until age 50 years.3,4,6 The timing hypothesis suggests that although estrogen may have neuroprotective effects when provided close to the menopausal transition, it can increase the risk of cognitive impairment or dementia if given to older women.3,20
Mood
The perimenopause has been described as a window of vulnerability for the development of both depressive symptoms and major depressive episodes.21 This concept suggests an increased sensitivity that some women may experience due to a fluctuating hormonal milieu.4,21 Studies have suggested a possible association between menopause staging and the risks for depressive symptoms or major depressive disorder (MDD). An overall 1.5-fold to 3.0-fold increased risk for depressive symptoms was observed during the menopausal transition. Additionally, this trend was noted in perimenopausal women with no history of depressive symptoms.4,21,22 Risk factors associated with development of depressive symptoms included demographic characteristics (age, race, education, financial problems), underlying health conditions, somatic symptoms (vasomotor symptoms, insomnia), psychosocial factors, high trait anxiety, and low social support.21,23 The majority of women who experience an MDD at midlife have experienced a prior depressive episode and may be at increased risk for recurrence. This is consistent with findings from the Study of Women’s Health Across the Nation (SWAN), which found that a major depressive event was less likely to occur in women with no history of a MDD.7,21
Women who undergo early BSO are at an increased risk for anxiety and depression compared with women who experience menopause naturally. Findings from the Mayo Clinic Cohort Study of Oophorectomy and Aging showed an increased risk of de novo depression and de novo anxiety symptoms in women following early BSO, with an increased risk identified in younger women at the time of surgery (median follow-up, 24 years).3,4,23
In perimenopausal women, estrogen therapy appears to have similar antidepressant effects to classic antidepressant agents used to treat depression. This effect, however, has not been demonstrated in the treatment of postmenopausal women, suggesting a window of opportunity for its use in the perimenopause.7,22
Sleep
Sleep disruption occurs in approximately one-third of women and worsens during the perimenopause.24 The incidence of insomnia is estimated at 39% to 60% in perimenopausal and postmenopausal women.25 During the perimenopause, women with vasomotor symptoms are more likely to experience disrupted sleep.7 This association was supported by findings from SWAN and the Penn Ovarian Aging Study.4 A subsample from the Korean Genome and Epidemiology Study also found that women who underwent surgical menopause were 2.1 times more likely to experience insomnia when compared with women experiencing natural menopause. Additionally, women in the surgical menopause group who engaged in sleep-interfering behaviors, such as drinking caffeine or alcohol, consuming large meals, or watching television before bed, had a greater severity of insomnia symptoms when compared with women in their referent group.4,25
Hormone therapy improves sleep in women with debilitating vasomotor symptoms. Some evidence suggests the beneficial effect of transdermal estrogen therapy on sleep in perimenopausal women, independent of vasomotor symptoms.7
Sexual function
Sexual dysfunction is one of the most prevalent and bothersome effects of early menopause. It is frequently multifactorial, related to the physiologic and psychological aspects of early hormonal deprivation.3,4,6,26 Other potential factors influencing sexual function include life-cycle stage, coping mechanisms, and family or societal influences.3
Sexual function is modulated through the effects of estrogens and androgens. Estrogens mediate peripheral arousal including vaginal congestion and lubrication. Androgens, primarily testosterone, have a central effect on neurotransmitters by promoting desire and arousal. Androgens also promote a peripheral response through an effect on nitric oxide.3,4 Premature or early menopause is associated with a gradual loss of ovarian function causing a progressive decline in estrogen, progesterone, and testosterone levels. Women with early BSO experience an abrupt and immediate hormonal decline resulting in more severe sexual dysfunction.4,17,23,26,27
Hypoactive sexual desire disorder (HSDD), defined as persistent deficiency or absence of sexual thoughts, fantasies, or desire with associated distress, is prevalent in women throughout the lifespan. HSDD is highest in women during midlife between ages 45 and 64 years. When compared to premenopausal women or women with natural menopause, higher rates of HSDD have been observed in women with early BSO.4
Genitourinary syndrome of menopause (GSM) is also prevalent in the postmenopausal population, affecting 50% of women.4,17 GSM is a chronic progressive condition negatively impacting sexual function, self-esteem, partner relationships, and quality of life. As with HSDD, the GSM symptom profile is more severe in women with early BSO, likely due to the abrupt loss of ovarian hormones.4,26,27 Similar findings have been noted in women with early menopause due to POI.4,6 Low-dose vaginal estrogen preparations have been recommended for the safe, effective treatment of GSM in the appropriate candidates.7,27 Although both systemic HT and low-dose vaginal estrogen therapy improve vaginal health, systemic therapy generally does not enhance sexual function independent of its effect on GSM. Transdermal estrogen has minimal effect on sex hormone-binding globulin and free testosterone levels and is the preferred treatment modality for symptomatic menopausal women with libido concerns.7
Management
There are no randomized clinical trials to date assessing the effect of HT on disease outcomes, but observational evidence suggests that HT lowers the risk of long-term morbidities associated with early hypoestrogenism. Limited observational studies also have shown no increased baseline risk of breast cancer in young BRCA-positive women using HT following risk- reducing BSO.7,28 Current clinical guidelines recommend the use of HT in young menopausal women (< 45 years) even in the absence of symptoms. The same exclusion criteria for HT use in women with natural menopause should be applied to this younger cohort. Treatment should be continued until the average age of menopause, at which time decisions regarding continued use should be reevaluated.2–4,7,9,11,13,16,26
Although precise data are lacking regarding serum estradiol levels needed to maintain cardiovascular and brain health, the use of higher estrogen doses are recommended to approach physiologic premenopausal levels.2–4,7–9,11,13,14,16,17 An estradiol level of 80 to 120 pg/mL, which approximates the average serum level of a menstruating woman, has been recommended. A significantly lower level reflects poor absorption, requiring a higher transdermal dose or a transition to oral therapy. A dose of 0.075 mg/day or 0.1 mg/day of transdermal estradiol (or a therapeutic equivalent) is suggested to achieve that range.3,9 Routine serum estradiol testing is generally not recommended unless symptoms persist with the use of higher-dose transdermal estrogen formulations. Clinicians, however, should be aware that serum monitoring may not be useful when using oral estrogens due to the inherent daily fluctuation associated with oral metabolism.9
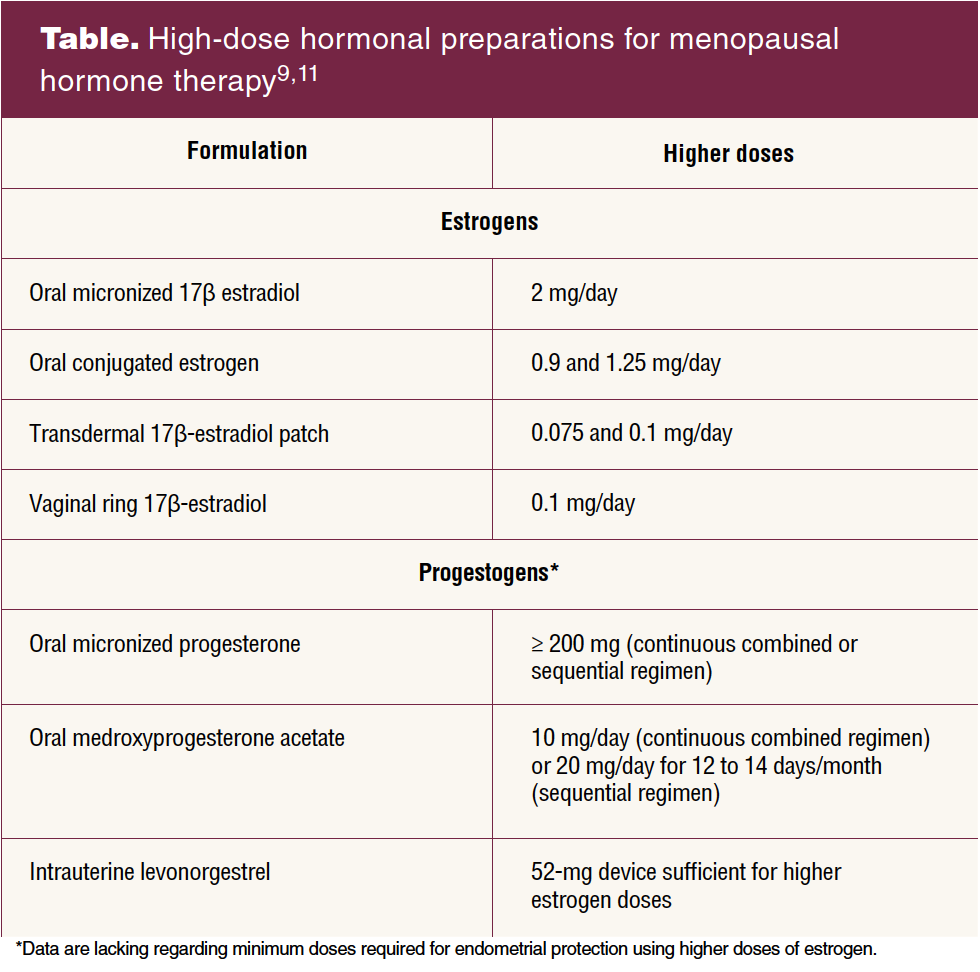
Replacement therapy can be achieved with the following estradiol preparations: oral micronized 17 β-estradiol 2 mg/day, oral conjugated estrogen 0.9 and 1.25 mg/day, transdermal 17 β-estradiol patch 0.075 mg and 0.1 mg/day, and the 17 β-estradiol vaginal ring 0.1 mg/day.
Transdermal and transvaginal estradiol preparations avoid the first pass effect and are recommended in patients at higher risk for cardiovascular or thromboembolic disease.13 In women with an intact uterus, the choice of estrogen therapy should be combined with the appropriate dose of progestogen for prevention of endometrial neoplasia.8,9 Data are lacking regarding minimum doses required with use of high-dose estrogen formulations. Current recommendations have been made based on expert opinion.9
Although both medroxypro-gesterone acetate and micronized progesterone improve cardiovascular risk markers including blood pressure and levels of high-density lipoprotein, LDL, and fibrinogen, recent evidence suggests that micronized progesterone may have a lower risk of cardiovascular events compared to other progestogens.13 Although combined oral contraceptives typically contain higher doses of estrogen, they can alternatively be used to treat younger women, particularly those with POI who require contraception.3 The Table lists higher-dose hormonal preparations for menopausal hormone replacement therapy (HRT).
It is important for clinicians to underscore the importance of HRT in mitigating disease risk and mortality rates in women with early estrogen loss. For those experiencing natural menopause, HT is approved for treatment of moderate-to-severe vasomotor symptoms, prevention of osteoporosis, and treatment of GSM (low-dose vaginal estrogen therapy is recommended in the absence of indications for systemic use).7 Although these clinical indications apply to women with early hypoestrogenism, higher doses are required to restore a premenopausal hormonal milieu for preservation of cardiovascular, brain, bone, and sexual health.2
The care of women with early menopause includes promotion of a healthy lifestyle including avoidance of tobacco, moderation of alcohol, regular aerobic exercise, and consumption of a healthy diet with the recommended daily intake of calcium and vitamin D.11,24,29 Current clinical guidelines for cervical, breast, and colorectal cancer screening should be followed.29 Bone density testing is recommended in women age 50 years and older with risk factors for low bone mass or fracture. Risk factors include low body weight (BMI < 21 kg/m2), history of prior fracture, family history of osteoporosis or hip fracture, current tobacco use, excessive alcohol intake, long-term use of high-risk medications such as glucocorticoids, and discontinuation of estrogen therapy.30 The use of statins also has been suggested for primary prevention of CVD in women with early menopause at intermediate risk (≥ 7.5% to < 20%).31
Implications for practice
Nurse practitioners have a pivotal role in the identification and treatment of women with early estrogen loss. The health consequences of early hypoestrogenism have been well established in the literature. Current clinical guidelines support the use of menopausal HT to obviate the health risks associated with early estrogen loss, even in the absence of bothersome symptoms. Efforts to increase patient education and promote public awareness are needed to disseminate accurate information regarding the health risks associated with early menopause and need for hormone replacement therapy in the appropriate candidates.
References
1 Crandall CJ, Bachman GA, Faubion SS, et al, eds. Menopause Practice: A Clinician’s Guide, 6th ed. Pepper Pike, OH: North American Menopause Society; 2019.
2 Faubion SS, Files JA, Rocca WA. When lowest dose for shortest amount of time does not apply. J Womens Health. 2016;25(4):416-417.
3 Faubion SS, Kuhle CL, Shuster LT, Rocca WA. Long-term health consequences of premature or early menopause and considerations for management. Climacteric. 2015;18(4):483-491.
4 Kingsberg SA, Larkin LC, Liu JH. Clinical effects of early or surgical menopause. Obstet Gynecol. 2020;135(4):853-868.
5 European Society for Human Reproduction and Embryology (ESHRE) Guideline Group on POI; Webber L, Davies M, Anderson R, et al. ESHRE guideline: management of women with premature ovarian insufficiency. Hum Reprod. 2016;31(5):926-937.
6 Panay N, Anderson RA, Nappi RE, et al. Premature ovarian insufficiency: an International Menopause Society white paper. Climacteric. 2020;23(5):426-446.
7 Faubion SS, Crandall CJ, Davis L, et al. The 2022 hormone therapy position statement of The North American Menopause Society. Menopause. 2022;29(7):767-794.
8 Committee on Gynecologic Practice. Hormone therapy in primary ovarian insufficiency. Obstet Gynecol. 2017;129(5):e134-e141.
9 Faubion SS, Kaunitz AM, Kapoor E. HT for women who have had BSO before the age of natural menopause: discerning the nuances. OBG Manag. 2022;34(2):20-27,45.
10 Rocca WA, Gazzuola Rocca L, Smith CY, et al. Loss of ovarian hormones and accelerated somatic and mental aging. Physiology. 2018;33(6):374-383.
11 Kaunitz AM, Kapoor E, Faubion S. Treatment of women after bilateral salpingo-oophorectomy performed prior to natural menopause. JAMA. 2021;326(14):1429-1430.
12 Szabo RA, Marino JL, Hickey M. Managing menopausal symptoms after cancer. Climacteric. 2019;22(6):572-578.
13 Sullivan SD, Sarrel PM, Nelson LM. Hormone replacement therapy in young women with primary ovarian insufficiency and early menopause. Fertil Steril. 2016;106(7):1588-1599.
14 Pinkerton JV. Hormone therapy for postmenopausal women. N Engl J Med. 2020;382(5):446-455.
15 Suzuki Y, Huang Y, Melamed A, et al. Use of estrogen therapy after surgical menopause in women who are premenopausal. Obstet Gynecol. 2022;139(5):756-763.
16 Rocca WA, Mielke MM, Gazzuola Rocca L, Stewart EA. Premature or early bilateral oophorectomy: a 2021 update. Climacteric. 2021;24(5):466-473.
17 Kaunitz AM, Faubion S. Surgical menopause: Health implications and hormonal management. Menopause. 2020;28(1):1-3.
18 Parker WH, Feskanich D, Broder MS, et al. Long-term mortality associated with oophorectomy compared with ovarian conservation in the Nurses’ Health Study. Obstet Gynecol. 2013;121(4):709-716.
19 Yoshida T, Takahashi K, Yamatani H et al. Impact of surgical menopause on lipid and bone metabolism. Climacteric. 2011;14(4):445-452.
20 Georgakis MK, Beskou-Kontou T, Theodoridis I, et al. Surgical menopause in association with cognitive function and risk of dementia: a systematic review and meta-analysis. Psychoneuroendocrinology. 2019;106:9-19.
21 Maki PM, Kornstein SG, Joffe H, et al; Board of Trustees for The North American Menopause Society (NAMS) and the Women and Mood Disorders Task Force of the National Network of Depression Centers. Guidelines for the evaluation and treatment of perimenopausal depression: summary and recommendations. Menopause. 2018;25(10):1069-1085.
22 Soares CN. Depression and menopause: an update on current knowledge and clinical management for this critical window. Med Clin North Am. 2019;103(4):651-667.
23 Rocca WA, Grossardt BR, Geda YE, et al. Long-term risk of depressive and anxiety symptoms after early bilateral oophorectomy. Menopause. 2008;15(6):1050-1059.
24 Pinkerton J. Knowledge gaps and challenges in care for menopausal women. OBG Manag. 2022;34(6):21-29.
25 Cho NY, Kim S, Nowakowski S, et al. Sleep disturbance in women who undergo surgical menopause compared with women who experience natural menopause. Menopause. 2019;26(4):357-364.
26 Larkin L, Kingsberg SA. The unintended sexual health consequences of risk-reducing surgery: we must do more! Menopause. 2019;26(2):118-120.
27 Faubion SS, Kingsberg SA, Clark AL, et al. The 2020 genitourinary syndrome of menopause position statement of the North American Menopause Society. Menopause. 2020;27(9):976-992.
28 Sarrel PM. A call to increase the use of hormone therapy to prevent disease in symptomatic postmenopausal women. Menopause. 2019;26(6):573-575.
29 Shifren JL, Gass MLS; NAMS Recommendations for Clinical Care of Midlife Women Working Group. The North American Menopause Society recommendations for clinical care of midlife women. Menopause. 2014;21(10):1038-1062.
30 North American Menopause Society Editorial Panel. Management of osteoporosis in postmenopausal women: the 2021 position statement of The North American Menopause Society. Menopause. 2021;28(9):973-997.
31 Cho L, Davis M, Elgendy I, et al. Summary of updated recommendations for primary prevention of cardiovascular disease in women: JACC state-of-the-art review. J Am Coll Cardiol. 2020;75(20):2602-2618.
Womens Healthcare. 2023;11(4):10-15,41. doi: 10.51256/WHC082310
Key words: early menopause, premature menopause, premature ovarian insufficiency, bilateral oophorectomy, estrogen, hormone replacement therapy