This pre/post-test study was undertaken to determine whether an algorithm constructed in an electronic health record template would aid in diagnosing interstitial cystitis. The algorithm is presented and the study methods, results, and implications are described here.
Interstitial cystitis (IC), also known as bladder pain syndrome, is a chronic illness with no definitive cause or cure. This disorder has no universally accepted name1 and is frequently underdiagnosed or misdiagnosed.2
Background information
 The American Urological Association (AUA) describes IC as a disorder manifested by a perceived sensation of pain and pressure related to the bladder and associated with lower urinary tract symptoms of greater than 6 weeks’ duration, in the absence of infection or other identifiable causes.3 The incidence of IC in the United States is 3%-6%.4 Like irritable bowel syndrome, chronic fatigue syndrome, and fibromyalgia, IC is a functional somatic syndrome with distressing symptoms that persist in the absence of structural pathology.1 IC is typically diagnosed in midlife,4 even though symptoms frequently arise in early childhood or adolescence.5,6 In fact, 25% of adults with IC, 90% of whom are female, report the existence of symptoms since they were children.7,8
The American Urological Association (AUA) describes IC as a disorder manifested by a perceived sensation of pain and pressure related to the bladder and associated with lower urinary tract symptoms of greater than 6 weeks’ duration, in the absence of infection or other identifiable causes.3 The incidence of IC in the United States is 3%-6%.4 Like irritable bowel syndrome, chronic fatigue syndrome, and fibromyalgia, IC is a functional somatic syndrome with distressing symptoms that persist in the absence of structural pathology.1 IC is typically diagnosed in midlife,4 even though symptoms frequently arise in early childhood or adolescence.5,6 In fact, 25% of adults with IC, 90% of whom are female, report the existence of symptoms since they were children.7,8
The etiology of IC is unclear and likely multifactorial. Proposed etiologies include neurogenic hypersensitivity, autoimmune deficiency, increased number and activation of mast cells containing inflammatory mediators, and a deficiency in the glycosaminoglycan (GAG) layer of the bladder.1 The GAG layer prevents toxins, acid, or infectious agents in urine from penetrating the bladder wall.1 When this protective layer breaks down, the bladder wall becomes irritated from exposure to urinary potassium. This irritation generates IC symptoms such as pelvic pain, urinary frequency/urgency, nocturia, and dyspareunia.9
Statement of the problem
In the U.S., among the 3.5-8 million women with IC symptoms, only 9.7% have received a diagnosis of IC.1 Healthcare providers’ (HCPs’) lack of knowledge about IC symptoms delays diagnosis and implementation of treatment.8 Without established diagnostic criteria, IC is identified based on the exclusion of other urogynecologic problems.9,10
Healthcare providers can follow the AUA’s guideline for diagnosis and treatment of IC.3 A portion of this guideline is derived from research and consensus-based processes, but IC diagnosis is based primarily on clinical principles and expert opinions.3 A consensus guideline for diagnosis and treatment was published by the Canadian Urological Association (CUA) in 2016.11 This guideline was based on Medline, PubMed, and consensus conference proceedings and categorizes what is mandatory, as well as recommended, for an IC diagnosis. Despite the availability of guidelines from the AUA and the CUA, not all HCPs are aware of them. Even if HCPs are familiar with the guidelines, they may not be using any kind of tool to facilitate the diagnostic process.
Making a diagnosis of IC
In the absence of established diagnostic criteria, how do HCPs diagnose IC in their patients?
Symptoms
For all patients with IC, pain is the hallmark symptom.3 The site of pain ranges from the umbilicus to the perineal, inguinal, or perirectal areas.12 Pain may also radiate down the legs.12 Pain during sexual intercourse, or dyspareunia, is reported by 60% of young patients with IC.1 Urinary frequency is often the first symptom of IC to arise, but patients may not recognize it as a problem until they have endured many years of it.5 Among patients with IC, 92% report urinary frequency: 86% say that they urinate more than 11 times during a 24-hour period and 71% say that they urinate 3 or more times at night—the definition of nocturia.1, 3
Differential diagnosis
The differential diagnosis (DDx) for IC appears in Table 1.8,13 Overactive bladder (OAB) and IC are often confused with each other because both conditions typically manifest with urinary frequency. The key differential symptom is pelvic pain; if such pain is present, OAB can be ruled out.14 Patients with OAB tend to void frequently because they fear leaking,8 whereas those with IC tend to void frequently—with small volume, regardless of whether it is day or night15—to relieve or avoid pain.16
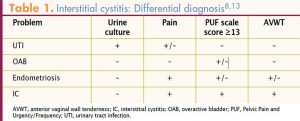
Endometriosis, with its presenting symptoms of pelvic pain and dyspareunia, may be included in the DDx for IC at first. Although pain associated with endometriosis may occur throughout the menstrual cycle, it usually worsens during menstruation. Pain associated with IC also occurs throughout the menstrual cycle but tends to worsen a few days before menses.11 Women with endometriosis may experience urinary urgency and frequency, but these symptoms are much less common than in women with IC. The physical examination finding of anterior vaginal wall tenderness (AVWT) points to IC, although it may be present in patients with endometriosis who have bladder involvement. AVWT can be determined by placing one finger in the vagina and palpating the anterior vaginal wall underlying the empty bladder. Finding AVWT on physical exam is 95% sensitive in correctly identifying IC.17 Another point to remember is that endometriosis and IC often coexist.18
Diagnostic tools
The Pelvic Pain and Urgency/Frequency (PUF) Patient Symptom Scale is a simple tool to further characterize a patient’s complaints and narrow the DDx. This scale includes urogynecologic symptoms and has three questions each on urinary frequency, urgency, and pelvic pain and two questions on symptoms associated with intercourse.5 A PUF scale score ≥13 justifies a possible IC diagnosis.11
A frequency volume chart (FVC) is a simple tool that assesses the average volume that a person is voiding. A person with IC voids an average of <100 mL 17-25 times a day, as compared with 289 mL 6 times a day in an asymptomatic person.11
A combination of negative urinalysis and urine culture findings, chronic urinary symptoms, chronic pelvic pain, and a positive AVWT finding on physical exam is sufficient evidence to diagnose IC. Adding an elevated PUF scale score and an FVC confirming frequent voiding in small amounts can give HCPs confidence that they have the correct diagnosis.
Knowledge-to-action model
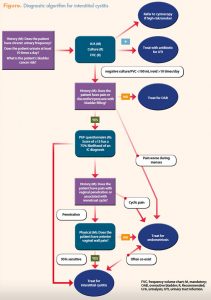
The knowledge-to-action (KTA) model provided a conceptual framework for the researcher to develop an evidence-based algorithm for documentation and diagnosis of chronic urinary symptoms, including IC. Using the KTA model, researchers and clinicians follow a sequence of phases in order to optimize knowledge translation across various fields of practice, with the goal of improving health outcomes.19 Knowledge tools/ products such as clinical practice guidelines, decision aids, and algorithms are developed after review of evidence pertinent to the topic.20 Once the authors accumulated and evaluated IC research and carefully reviewed evidence-based guidelines, a diagnostic tool was developed using an algorithm format (Figure). A Centricity electronic health record (EHR) template was constructed from the algorithm.
The project
Purpose
The purpose of this project was two-fold. The first aim was to develop a diagnostic tool—that is, an EHR algorithm template called Chronic Urinary Symptoms—that was based on available evidence to assist HCPs in identifying women with IC. The second aim was to study outcomes resulting from the use of this tool, particularly in terms of HCPs’ satisfaction with it, their perceived competence in diagnosing IC, and their increased inclusion of IC in the DDx for chronic urinary symptoms.
Methods
Design
A pre-test/post-test quasi-experimental design was used to answer these questions: Will HCPs report satisfaction with the use of an HER algorithm template for chronic urinary symptoms? Will HCPs experience competence in diagnosing IC when using an EHR algorithm template for chronic urinary symptoms? Will HCPs consider IC more often in their DDx when using an EHR algorithm template for chronic urinary symptoms? The statistical package used throughout the study was IBM SPSS Statistics Version 23.0.
Setting and participants
The project took place at a federally qualified health center (FQHC) and included a convenience sample of HCPs in women’s health, internal medicine, and family practice. Inclusion criteria were (1) HCPs who saw female patients aged 16 years or older as a primary care provider or for their female healthcare needs and (2) use of the Centricity EHR. Approval was given by the Institutional Review Board of Edinboro University of Pennsylvania and the Clinical Issues Committee of the FQHC.
Among 53 HCPs who met inclusion criteria, 19 agreed to participate in the project and gave signed informed consent. Using a PowerPoint presentation, the project leader met with each participant individually to provide education on both IC and the operation of the Chronic Urinary Symptoms template on the EHR.
Data collection and tools
Participants completed a modified Post-Study System Usability Questionnaire (PSSUQ) and a Perceived Competence Scale (PCS) questionnaire at the completion of the 4-month trial. Questionnaires were returned by inter-office mail, with no identifiers on them, to maintain confidentiality. In addition, use of 21 common bladder ICD-10 codes, including IC with or without hematuria, was compared 4 months before and 4 months after participants were introduced to the EHR algorithm template. For this comparison, a unique identifier designating each participant was given by the clinical program analyst, who assessed the ICD-10 diagnostic habits for each participant.
The PSSUQ is a 16-item survey that calculates satisfaction with a product—in this case, the Chronic Urinary Symptoms template on the EHR.21 Participants responded to each item on a 7-point Likert-type scale from 1 (strongly agree) to 7 (strongly disagree). Averaging the subscales of System Quality, Information Quality, and Interface Quality gave an overall satisfaction score that was highly reliable (α = .94).22 A comments section and an opportunity for participants to make suggestions to improve the template were added to the questionnaire.
The PCS is a 4-item questionnaire assessing a person’s perceived competence in a particular subject.23 The scale was used to measure overall perception of competence in diagnosing IC. Participants responded to each item on a 7-point Likert-type scale from 1 (strongly disagree) to 4 (neutral) to 7 (strongly agree). The α measure of internal consistency of the PCS was reported as >.80. The questionnaire is copyrighted and can be used free of charge for academic research projects.23 Participants who answered affirmatively to this question—Did you consider IC as a differential diagnosis on any patient in the past 4 months?— received a follow-up question: If you have considered IC more often, did you use the ICD-10 code for interstitial cystitis?
Results
A total of 19 HCPs agreed to participate and 14 returned the questionnaires, a response rate of 73.7%. The sample included 12 physicians (63%), 4 nurse practitioners (21%), and 3 physician assistants (16%), with 13 (68%) in family practice, 4 (21%) in women’s health, and 2 (11%) in internal medicine. Mean number of years of practice was 12.9 (range, 1-38) and mean number of years of using an EHR was 4.7 (range, 1-10). All but one participant felt “good” to “extremely comfortable” with use of the EHR.
Participants “moderately agreed” that the algorithm provided the necessary information to manage patients with chronic urinary symptoms, with a mean (M) score of 2.036 (standard deviation [SD], 1.081), and that the template was usable. Participants scored Information Quality (M, 1.92; SD, .97) of the template better than System Quality (M, 2.04; SD, 1.11) or Template Quality (M, 2.07; SD, 1.17). Information Quality entailed clarity and organization of the material and ease of finding items of information. System Quality evaluated the ease in learning and using the template. Template Quality measured the functions, capabilities, graphics, and language of the program. Even though participants felt the Information Quality subscale exceeded the other subscales, a one-way repeated measure ANOVA found no difference in the scores.
Given the relatively high average scores on the PCS questionnaire (Table 2), participants perceived that they were competent in diagnosing IC. They noted that, using the template, they would consider IC more often as a diagnosis. With regard to the question about actually using the ICD-10 code for IC, 6 participants did use it, 5 did not, and 3 did not answer this question.
ICD-10 codes evaluated were used a total of 235 times during the 4 months prior to implementation of the template and a total of 290 times during the 4 months after implementation. A Wilcoxon signedrank test determined that there was a significant median increase in the total number of IC diagnoses from before (n = 0) to after (n = 5) implementation of the EHR template (z = 2.236; P = .025).
Discussion
Success of the project was determined by comparing pre- and post-ICD-10 codes used and participants’ assessment of the usability of the algorithm. Results of the study of outcomes for the project indicated that the EHR algorithm template did assist participants in diagnosing IC.
Diagnosis of IC before and after implementation of HER algorithm template
Of the 19 participants, 5 increased their diagnosis of IC following introduction of the template; there were no IC diagnoses at either time point for the other 14 participants. Although ICD-10 codes for IC with/without hematuria were not used by every participant, all but 1 did utilize the template at least once for chronic urinary symptoms during the study time frame. That is, participants at least considered IC in the DDx.
The 5 participants who denied using the ICD-10 code for IC with/without hematuria had a lower average competence score (M, 5.86; SD, .29) than the 6 who did use the ICD-10 code for IC (M, 6.37; SD, .56), but the difference was not significant. The fact that the groups did not differ significantly in this regard is not surprising because the sample was very small and the interpretation must be explained with caution.
Experience with the EHR algorithm template
Mean satisfaction scores for each question on the PSSUQ revealed where improvements could be made to the template. Poorest scores were in functions and capabilities (M, 2.08; SD, 1.04) of the Template Quality subscale, and the potential to quickly be productive (M, 2.57; SD, 1.60) and recovering from mistakes easily and quickly (M, 2.36; SD, 1.36) in the System Quality subscale. Participants offered only a few comments, which led to improvements in the Template Quality subscale.
Limitations
The template made for this project is specific to the Centricity EHR system and may not be widely generalizable to other systems. Evaluation time was short and the sample size was small. Ensuring participants’ confidentiality resulted in the inability to assess a connection between participants’ answers to the surveys and the diagnosis codes used, which made it impossible to know whether anything about the usability would effectuate a change in diagnosis.
Clinical implications
Interstitial cystitis is a poorly understood disorder. Diagnosis is often delayed and difficult because no objective signs or tests have been established to confirm it.23 Patients who experience chronic urinary symptoms but receive no diagnosis may experience adverse physical and psychological effects. Giving HCPs an accepted evidence-based diagnostic algorithm template for chronic urinary symptoms may increase their confidence in diagnosing and treating IC. The project succeeded in providing a foundation for developing an easy-to-follow algorithm to guide HCPs in correctly diagnosing chronic urinary problems. Although the sample size was small and adjustments to the algorithm were recommended, it is a good start for developing other templates in other EHR systems. This project demonstrated that a well-formulated diagnostic algorithm template can assist HCPs in differentiating IC from other urogynecologic conditions. Developing an application for a smartphone or personal computer could make the algorithm more readily available to HCPs.
Anita L. Booth is a nurse practitioner at Primary Health Network, a federally qualified health center in Pennsylvania. She has specialized in women’s health for the past 16 years and has been working with patients with interstitial cystitis for the past decade. She can be contacted at ABooth@primary-health.net. Jill Harpst Rodgers is Assistant Professor of Nursing and Co-Coordinator of the DNP Program at Edinboro University in Edinboro, Pennsylvania. The authors state that they do not have a financial interest in or other relationship with any commercial product named in this article.
References
1. McLennan MT. Interstitial cystitis: epidemiology, pathophysiology, and clinical presentation. Obstet Gynecol Clin North Am. 2014;41(3):385-395.
2. Holroyd S. Diagnosis and treatment of painful bladder syndrome. J Commun Health Nurs. 2016;30(6):48-53.
3. Hanno PM, Erickson D, Moldwin R, et al. Diagnosis and treatment of interstitial cystitis/bladder pain syndrome: AUA guideline amendment. J Urol. 2015;193(5):1545-1553.
4. Mckernan LC, Nash M, Carr ER. An integrative trauma-based approach toward chronic pain: specific applications to interstitial cystitis/bladder pain syndrome. J Psychother Integr. 2016;26(3):1-17.
5. Parsons CL. Diagnosing the bladder as the source of pelvic pain: successful treatment for adults and children. Pain Manag. 2014;4(4):293-301.
6. Hammett J, Krupski TL, Corbett ST, et al. Adolescent pelvic pain: interstitial cystitis. J Pediatr Urol. 2013;9(3):134-137.
7. Davis NF, Gnanappiragasam S, Thornhill JA. Interstitial cystitis/painful bladder syndrome: the influence of modern diagnostic criteria on epidemiology and on internet search activity by the public. Transl Androl Urol. 2015;4(5):506-511.
8. Whitmore KE, Theoharides TC. When to suspect interstitial cystitis. J Fam Pract. 2011;60(6):340-347.
9. Jiang YH, Jhang JF, Kuo HC. Revisiting the role of potassium sensitivity testing and cystoscopic hydrodistention for the diagnosis of interstitial cystitis. PLoS One. 2016;11(3):1-9.
10. Bosch PC, Bosch DC. Treating interstitial cystitis/bladder pain syndrome as a chronic disease. Rev Urol. 2014;16(2):83-87.
11. Cox A, Golda N, Nadeau G, et al. CUA guideline: diagnosis and treatment of interstitial cystitis/bladder pain syndrome. Can Urol Assoc. 2016;10(5-6):136-155.
12. Killinger KA, Boura JA, Peters KM. Pain in interstitial cystitis/bladder pain syndrome: do characteristics differ in ulcerative and non-ulcerative subtypes? Int Urogynecol J. 2013;24(8):1295-1301.
13. Warren JW, Wesselmann U, Greenberg P, et al. Urinary symptoms as a prodrome of bladder pain syndrome interstitial cystitis. J Urol. 2014;83(5):1035-1040.
14. Barr S. Diagnosis and management of interstitial cystitis. Obstet Gynecol Clin North Am. 2014;41(3):397-407.
15. Kim SH, Oh SA, Oh SJ. Voiding diary might serve as a useful tool to understand differences between bladder pain syndrome/interstitial cystitis and overactive bladder. Int J Urol. 2014;21(2):179-183.
16. Homma Y. Hypersensitive bladder: a solution to confused terminology and ignorance concerning interstitial cystitis. Int J Urol. 2014;21(1):43-47.
17. Paulson JD, Paulson JN. Anterior vaginal wall tenderness (AVWT) as a physical symptom inchronic pelvic pain. JSLS. 2011;15(1):6-9.
18. Butrick CW, Howard FM, Sand PK. Diagnosis and treatment of interstitial cystitis/painful bladder syndrome: a review. J Womens Health. 2010;19(6):1185-1193.
19. Graham ID, Logan J, Harrison MB, et al. Lost in knowledge translation: time for a map? J Contin Educ Health Prof. 2006;26(1):13-24.
20. Kastner M, Straus SE. Application of the knowledge-to-action and medical research council frameworks in the development of an osteoporosis clinic decision support tool. J Clin Epidemiol. 2012;65(11):1163-1170.
21. Garcia A. UX Research: Standardized Usability Questionnaire. November 27, 2013.
22. Deci EL, Ryan RM. Self-determination theory. Perceived Competence Scales. 2016.
23. Berry SH, Elliott MN, Suttorp M, et al. Prevalence of symptoms of bladder pain syndrome/interstitial cystitis among adult females in the United States. J Urol. 2011;186(2):540-544.
“]

