Before reading the article, click here to take the pretest.
Overactive bladder (OAB) is a common condition affecting women in every age bracket. OAB symptoms need no longer undermine a woman’s quality of life. After ascertaining a patient’s goals, a healthcare provider can devise an individualized treatment plan that may include simple lifestyle changes, an FDA-approved medication regimen, and nonpharmacologic interventions such as pelvic floor muscle exercises, bladder training, and use of a pessary.
Key words: overactive bladder, OAB, urinary incontinence, nocturia, anticholinergics, beta-3 adrenergic agonist
Anita Padd, age 69, is a retired registered nurse who presents with a list of urinary complaints that she attributes to the fact that, when she was working, she was too busy to take time to go to the bathroom. A week before her appointment, Anita had attended a luncheon sponsored by a nurse practitioner (NP) who recently opened a pelvic health center around the corner. Anita says that she is glad both that she attended the luncheon—“For the first time in years, I realized that I am not alone and that help is available for my bladder problems”—and that she has made an appointment to see this NP.
Why are so many women experiencing bladder problems now?
Anita, born in 1946, was among the first Baby Boomers to turn 65. She, like many of her peers, is healthier than members of previous generations and has lived long enough to develop one or more chronic health conditions such as hypertension, diabetes, and overactive bladder (OAB). According to the U.S. Census Bureau, in 2014 there were 76.4 million Baby Boomers—that is, persons born between 1946 and 1964.1 The number and proportion of elderly persons in the country will keep rising for many years to come.
Anita believes that her bladder problem is a normal consequence of aging. She has been managing the problem by restricting her fluid intake and wearing pads. She tells the NP that she didn’t broach the topic to her primary care provider (PCP) because she was embarrassed and because she feared that surgery was the only treatment available.
Are Anita’s perceptions/concerns about her bladder problem common?
Results of a 2012 nationwide survey of women aged 40-65 years showed that those who had mild to moderate OAB symptoms (n = 652), as compared with those who had no OAB symptoms (n = 1,017), tended to feel much less “in charge” of their lives and their health and were much more fearful of public embarrassment related to not being able to reach a toilet in time to avoid having an accident.2 OAB sufferers were also more likely to think that bladder problems, like wrinkles and gray hair, were a normal part of aging.
Anita’s PCP has never screened her for urinary problems. Like many PCPs, she manages hundreds of patients with multiple conditions and feels that investigating a condition such as OAB would be “opening Pandora’s box.” Like her patient, this PCP is not aware of conservative treatments for bladder problems and is confused by all the medications on the market.
How common is OAB? Should healthcare providers (HCPs) be routinely screening their patients for it?
The prevalence of OAB among women is 15%-17%.3,4 In raw numbers, at least 11-16 million women in the United States have OAB symptoms.4 Most women with OAB symptoms do not talk with their HCPs about their urinary dysfunction, and providers may not routinely inquire about it. As a result, only a small minority of women with OAB receive treatment for it.4
Healthcare providers who see women on a regular basis in their practice can fill the gap by routinely screening for OAB and by gaining the knowledge to treat it appropriately. In an NPWH member survey of 300 NPs that was conducted in 2013, 48.3% of respondents reported being confident in their ability to diagnose OAB and 41.1% felt sure of themselves with regard to treating OAB, but only 28.5% thought that most OAB sufferers in their practice had been identified (H.A.C., unpublished NPWH member survey data, 2013). With proper screening for OAB, most, if not all, of the patients with this condition can be identified and treated.
What exactly is overactive bladder?
Overactive bladder is a symptom complex consisting of urgency, frequency, and nocturia and, in 37% of cases, urge incontinence. Sudden and compelling urinary urgency, the hallmark of OAB, occurs as a result of premature and independent contractions of the bladder that escape inhibition by the central nervous system. Urinary frequency is defined as voiding 8 or more times in a 24-hour period.5 Nocturia entails awakening more than 2 times a night to urinate.5 For OAB sufferers who have urge incontinence, urine loss varies from a few drops to a “gush.”
The three main categories of urinary incontinence are urge incontinence, stress incontinence, and mixed incontinence. Urge incontinence is an involuntary loss of urine immediately preceded by or synchronous with a strong urge to void. Urine loss can be substantial, because bladder contractions may continue until the bladder is empty. Urge incontinence, also described as detrusor instability or detrusor hyperactivity,6 differs from a normal strong urge to void that can be controlled. It is due to spontaneous bladder spasm, which can result from dietary factors (bladder stimulants such as caffeine or alcohol), increased fluid intake, side effects of medication (e.g., diuretics, bethanechol), urinary tract infection/cancer, or nerve dysfunction associated with nerve trauma, diabetes, multiple sclerosis, or spinal cord injury.5,6 Many episodes of urge incontinence have triggers such as unlocking the door to one’s house upon return (key-in-lock syndrome), approaching a toilet, or hearing the sound of running water.7Stress incontinence is marked by an involuntary loss of urine due to increased abdominal pressure on the bladder that exceeds maximal urethral closure pressure.8 An episode of stress incontinence is precipitated by an activity such as coughing, sneezing, laughing, lifting, stepping off a curb, or tripping. Stress incontinence is due to an increase in abdominal forces in the presence of an anatomic weakness of the bladder neck, which typically maintains the seal of urine during activity. It can stem from a variety of situations (e.g., coughing) or conditions such as vaginal delivery, aging, estrogen deficiency associated with menopause, or obesity.6 Stress incontinence differs from urge incontinence in that it (1) is rarely associated with nocturia or an urge to urinate; (2) is precipitated by an activity such as coughing; and (3) often occurs at unexpected or inappropriate times.
Mixed incontinence is a combination of stress incontinence and urge incontinence. It is described as stress dominant or urge dominant. Its occurrence increases with aging.
Anita sits on the examination table at the NP’s office. Her complaints include (1) severe urgency to urinate when she arrives at home; (2) urinary frequency in the late morning; (3) nocturia: she needs to urinate 3-4 times a night, although she is thankful that she can usually go back to sleep; (4) urine leaking with position change when her bladder is full; and (5) feeling exhausted all the time. Anita indicates that she took hormone therapy years ago, but not recently. She reports vaginal dryness and long-standing constipation. She also reports that she takes cranberry pills because they are “good for the bladder.”
What does the workup for a patient with suspected OAB entail?
In its most recent guideline. the American Urological Association (AUA) lists these clinical diagnostic principles:
- Minimum requirements are a careful history, physical examination, and urinalysis.
- If deemed necessary, a urine culture and/or post-void residual assessment may be performed and information from bladder diaries and/or symptom questionnaires may be obtained.
- Urodynamics, cystoscopy, and diagnostic renal and bladder ultrasound are not needed in the initial workup of a patient with an uncomplicated presentation.9
History
The history includes a general health history and a focused history regarding lower urinary tract symptoms, including their onset, nature, duration, severity, and effect on quality of life.5 The HCP inquires about the presence or absence of diabetes, neurologic disorders, recurrent urinary tract infection, hematuria, kidney stones, previous lower abdominal or pelvic surgery, pelvic organ prolapse (POP), and vaginitis.10 In addition, the HCP asks the patient about her use of prescription and over-the-counter medications, particularly with regard to anticholinergics or antimuscarinics, antidepressants, antipsychotics, sedatives or hypnotics, diuretics, caffeine, alcohol, opioids, alpha-adrenergic blockers, alpha-adrenergic agonists, beta-adrenergic agonists, and calcium channel blockers.5 The HCP then poses screening questions specific to OAB:
- Do you ever leak urine when you have a strong urge on the way to the bathroom? How often?
- How frequently do you urinate during the day?
- How many times do you get up to urinate after going to sleep? Is it the urge to urinate that wakes you?
- How many pads a day do you wear for protection?
- Does this problem inhibit any activity or prevent you from doing things you like to do?
Physical examination
A comprehensive physical examination for OAB includes a pulmonary and cardiovascular evaluation and neurologic, abdominal, pelvic, and rectal exams.9,10 The pelvic exam can reveal findings such as genitourinary syndrome of menopause (GSM; also known as urogenital atrophy, vulvovaginal atrophy, and atrophic vaginitis) or POP, which can cause or exacerbate urinary symptoms. Assessment of pelvic muscle tone is done by inserting 1 or 2 fingers 2 cm into the patient’s vagina, palpating at 5 and 7 o’clock, asking the patient to tighten her rectal muscles, comparing the contralateral sides, noting her muscle strength and endurance (i.e., her ability to hold for 10 seconds), monitoring for inappropriate use of accessory muscles (e.g., abdominal or gluteal muscles), and encouraging the patient to relax her abdominal muscles.
Bladder diary
If a patient answers affirmatively to any of the screening questions posed during the history, she is asked to complete a bladder diary that will be reviewed during a subsequent visit. This diagnostic tool shows a woman’s day-to-day bladder habits and voiding patterns. She is asked to document the time, type, and amount of fluid intake (the type of fluid can indicate whether she is ingesting bladder irritants), the time of each void, each accidental leaking, and a notation of the volume of urine loss in subjective terms: large (>¼ cup), medium (<¼ cup), or small (dribbles).
Readers can access a daily bladder diary from the National Institute of Diabetes and Digestive and Kidney Diseases.11 A 3-day bladder diary is ideal.5 To evaluate the data in the bladder diary, HCPs need to be familiar with normal voiding values, which are as follows:
- Mean 24-hour urinary output (both men and women): 1,700 mL
- Mean number of daily voids:
6 to 7 - Mean bladder capacity: 330 mL (individual patients vary considerably; 300-500 mL is considered the normal range).<supP12
Vaginal pH
Without estrogen, the pH of vaginal secretions changes and the normal discharge becomes more alkaline (usually above 4.5), which is due to a decrease in vaginal lactobacilli. These changes cause vaginal tissues to thin, which can have adverse implications for the urogenital tract. Vaginal pH (normal range, 3.5-4.5) can be easily and inexpensively measured in the office by collecting a sample from the upper lateral vaginal wall and using litmus paper for pH testing.
Anita’s bladder diary indicates that, despite her best intentions, she is ingesting a host of bladder irritants, including the aforementioned cranberry pills, orange juice, green tea, and lemon seltzer. She voids 11-12 times a day and 3-4 times a night, and feels the urge to void even after she has just emptied her bladder. The exam shows that Anita has weak pelvic muscle contractions, a grade 2 cystocele, and GSM related to long-term estrogen deficiency. Her urinalysis results are negative or normal and her vaginal pH is in the alkaline range.
How is OAB identified?
The condition is identified by the presence of urinary urgency and frequency, nocturia and, in more than one-third of cases, incontinence.
Based on findings from the workup, Anita’s NP concludes that she has OAB. In addition, Anita meets diagnostic criteria for mixed urinary incontinence, urge dominant; urinary frequency; pelvic floor muscle weakness; urogenital atrophy; and chronic constipation.
What are the goals of OAB treatment?
Treatment goals are individualized and may vary from symptomatic improvement to complete symptomatic relief of urgency, frequency, nocturia, and urge incontinence. Asking patients with bladder control problems about their goals before treatment begins is useful in determining how aggressive the treatment should be.
At her initial appointment, although Anita is experiencing symptoms of both urge incontinence and stress incontinence, she reports that her most bothersome complaint is the nocturia. She states that she is tired all day as a result of awakening 3-4 times per night to urinate. She has even ceased participating in certain social activities because of her fatigue. When asked about her goals for treatment, Anita asserts that she does not want surgery or “anything invasive,” and that she would be delighted if she could get more sleep and was not always rushing to get to the bathroom. She and her HCP agree that the initial phase of treatment will focus on the urge aspect of her mixed incontinence symptoms.
What constitutes first-line treatment for OAB?
Many nonpharmacologic and pharmacologic approaches are available as first-line treatment for OAB.
Lifestyle changes
One of the simplest lifestyle changes is to avoid ingesting bladder irritants, many of which appear on the Carcio “C” List (Figure). Another easy approach is to manage fluid intake. The amount of fluid intake recommended depends on each patient’s body size, temperature, and physical activity level. In general, though, most women should aim to consume 4-8 cups of fluid (water plus all other liquids) a day. They should limit the amount of fluid ingested past 6 PM (or within 5 hours of the time when they plan to go to sleep). If they are excreting pale yellow urine, they are neither under-hydrating nor over-hydrating.

Pharmacotherapy
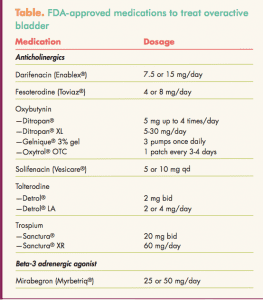
Two different classes of drugs, anticholinergics (antimuscarinics) and a beta-3 adrenergic agonist, are approved by the FDA for the treatment of OAB with symptoms of urge incontinence, urgency, and frequency (Table). The Sidebar lists pearls to prescribing these agents.
Anticholinergics work by blocking the effects of acetylcholine at muscarinic receptors in the bladder, thereby inhibiting involuntary detrusor contractions and reducing urgency.10 Side effects such as dry mouth, constipation, and blurred vision may be bothersome enough to prompt treatment discontinuation. Side effects are generally milder with extended-release formulations. Initiating the use of artificial saliva mouthwash and instituting measures to control constipation may enhance compliance with the regimen. All of the anticholinergics have similar efficacy in clinical trials; the key is finding a particular product that an individual patient can tolerate.
The beta-3 adrenergic agonist relaxes the detrusor smooth muscle during the storage phase of the urinary bladder fill-void cycle by activation of beta-3 adrenergic receptors, which increases bladder capacity.13 This agent can increase blood pressure.
Other approaches to management
The AUA supports the use of behavioral therapies such as bladder training, bladder control strategies (e.g., urge suppression), pelvic floor muscle training (PFMT; also known as Kegel exercises), and fluid management as first-line therapy for all patients with OAB.9 Studies have suggested that PFMT, alone or combined with biofeedback training or electrical stimulation, may be effective for treating OAB.14-16 Avoidance of constipation, which can further weaken the pelvic floor as a result of chronic straining, is another strategy.17 Increasing the amount of dietary fiber to 30 g/day can help prevent constipation.
A vaginal pessary, a flexible device made of silicone indicated for the treatment of POP or stress urinary incontinence,17,18 can be used to treat symptoms of OAB. A retrospective parallel cohort study was conducted on women whose OAB was treated with a ring pessary or multi-component behavioral therapy (MCBT) over a 42-month period.19 The ring pessary and MCBT had similar cure rates (29 of 150 [19%] vs. 46 of 231 [20%], respectively; P = .889), regardless of whether women were premenopausal (4 of 31 [13%] vs. 14 of 68 [21%], P?=?.358) or postmenopausal (25 of 119 [21%] vs. 32 of 163 [20%]; P?=?.776). Pessaries are usually fitted by an HCP and require a prescription, but a new over-the-counter pessary is also available.
Vaginal estrogen, indicated for treatment of the symptoms of GSM, may help relieve symptoms of OAB. The epithelial linings of the vagina and urethra have the highest concentration of estrogen receptors in the body and are therefore highly sensitive to alterations in estrogen levels.20 The estradiol vaginal ring (Estring®), which works particularly well in older women, is changed every 3 months. It can be used in conjunction with a pessary, which is also changed every 3 months. Vaginal estrogen cream products include estradiol cream (Estrace® Cream) and conjugated equine estrogens cream (Premarin® Vaginal Cream), which are inserted with an applicator. These creams are considered messy by some women, but they do add lubrication. Estradiol vaginal tablets (Vagifem®), less messy than the creams, are a good choice for women with a stenotic introitus because of the smaller applicator.
Anita returns to the NP’s office after 3 weeks. She reports that she has modified her diet to decrease her intake of bladder irritants. She drinks an adequate amount of fluids each day but limits her fluid intake, with sips only, after 6 pm. She takes a daily medication to treat her OAB, and denies any dry mouth, constipation, or drowsiness. She uses a pessary, which she finds comfortable, and inserts an estradiol vaginal ring every 3 months. She takes a stool softener to reduce constipation. She has attended two pelvic floor rehabilitation sessions and does Kegel exercises at least twice daily, and she can now wait up to 2.5 hours in between voids. She needs to urinate only twice during the night and rarely experiences a leak with position changes. Although Anita wears a pad only when she goes out, for “insurance,” she really doesn’t need it any longer. As a result of this multifaceted therapeutic approach, her urinary urgency and frequency, nocturia, leaks, fatigue, and worries have all diminished.
Conclusion
Women’s healthcare providers can routinely screen their patients for symptoms of OAB. In those patients who screen positive and in whom other causes of the symptoms have been excluded, a tool such as a bladder diary can be used to identify OAB. HCPs can then educate patients about avoidance of bladder irritants and about techniques to strengthen their pelvic floor muscles. If these measures are unsuccessful, HCPs can prescribe an FDA-approved medication to further alleviate symptoms. In all cases, a treatment approach based on a woman’s own needs and goals is most likely to be successful.
References
1. Population Reference Bureau. Just How Many Baby Boomers Are There? April 2014. prb.org/Publications/
Articles/2002/JustHowManyBabyBoomersAreThere.aspx
2. Muller N. Anxiety and fears in women with overactive bladder. Ostomy Wound Manag. January 2013. .o-wm.com/files/owm/pdfs/OWM_January2013_Muller.pdf
3. Stewart WF, Van Rooyen JB, Cundiff GW, et al. Prevalence and burden of overactive bladder in the United States. World J Urol. 2003;20(6):327-336.
4. Hartmann KE, McPheeters ML, Biller DH, et al. Treatment of overactive bladder in women. Evidence Report/Technology Assessment No. 187. 2009. ahrq.gov/clinic/tp/bladdertp.htm
5. Ellsworth PI. Overactive bladder. Medscape Drugs and Diseases. Updated April 25, 2014. emedicine.
medscape.com/article/459340-overview#aw2aab6b2b2
6. Brigham and Women’s Hospital website. Types of Incontinence and Risk Factors. Last modified on August 22, 2014. brighamandwomens.org/departments_and_services/obgyn/services/urogynecology/incontoverview.aspx
7. Carcio HA. Calming the overactive bladder: a nurse practitioner perspective. Womens Healthcare. 2014;
2(3):25-26, 49.
8. Tanagho EA, Bella AJ, Lue TF. Urinary incontinence. In: Tanagho EA, McAninch JW, eds. Smith’s General Urology. 17th ed. New York, NY: McGraw-Hill Medical. 2008:473-489.
9. Gormley EA, Lightner DJ, Burgio KL, et al; American Urological Association; Society of Urodynamics, Female Pelvic Medicine & Urogenital Reconstruction. Diagnosis and treatment of overactive bladder (non-neurogenic) in adults: AUA/SUFU guideline. J Urol. 2012;188(6 suppl):2455-2463.
10. Association of Reproductive Health Professionals. Diagnosis and Management of Overactive Bladder. April 2011. arhp.org/Publications-and-Resources/Quick-Reference-Guide-for-Clinicians/OAB/Diagnosis
11. National Institute of Diabetes and Digestive and Kidney Diseases. Your Daily Bladder Diary. niddk.nih.gov/
health-information/health-topics/urologic-disease/daily-bladder-diary/Documents/diary_508.pdf
12. Parsons M, Amundsen CL, Cardozo L, et al. Bladder diary patterns in detrusor overactivity and urodynamic stress incontinence. Neurourol Urodyn. 2007;26(6):800-806.
13. Andersson KE, Martin N, Nitti V. Selective ?3-adrenoceptor agonists for the treatment of overactive bladder. J Urol. 2013;190(4):1173-1180.
14. Burgio KL. Update on behavioral and physical therapies for incontinence and overactive bladder: the role of pelvic floor muscle training. Curr Urol Rep. 2013;14(5):457-464.
15. Shamliyan TA, Kane RL, Wyman J, Wilt TJ. Systematic review: randomized, controlled trials of nonsurgical treatments for urinary incontinence in women. Ann Intern Med. 2008;148(6):459-473.
16. Liaw Y-M, Kuo H-C. Biofeedback pelvic floor muscle training for voiding dysfunction and overactive bladder. Incont Pelvic Floor Dysfunct. 2007;1:13-15.
17. Lukacz ES. Patient information: Urinary incontinence treatments for women (Beyond the Basics). UptoDate. Last updated May 11, 2015. uptodate.com/contents/urinary-incontinence-treatments-for-women-beyond-the-basics
18. Clemons JL. Vaginal pessary treatment of prolapse and incontinence. UptoDate. Last updated March 20, 2014. uptodate.com/contents/vaginal-pessary-treatment-of-prolapse-and-incontinence
19. Sze EH, Hobbs G. A retrospective comparison of ring pessary and multicomponent behavioral therapy in managing overactive bladder. Int Urogynecol J. 2014;25(11):1583-1588.
20. Brincat M, Muscat Baron Y, Galea R, Buhagiar A. Estrogen deficiency and connective tissues. In: Crosignani PG, Paoletti R, Sarrel PM, et al, eds. Women’s Health in Menopause: Behaviour, Cancer, Cardiovascular Disease, Hormone Replacement Therapy. Springer Science+Business Media Dordrecht; 1994.
For readers of the online issue who wish to participate in this CE program, click here.